All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a Healthcare Professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The Lymphoma Hub uses cookies on this website. They help us give you the best online experience. By continuing to use our website without changing your cookie settings, you agree to our use of cookies in accordance with our updated Cookie Policy
Introducing

Now you can personalise
your Lymphoma Hub experience!
Bookmark content to read later
Select your specific areas of interest
View content recommended for you
Find out moreThe Lymphoma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Lymphoma Hub cannot guarantee the accuracy of translated content. The Lymphoma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
AACR 2017 | Targeting BCL2 and MCL1 in blood cancers
Bookmark this article
On April 5th 2017, during this year’s American Association for Cancer Research (AACR) annual meeting, Andrew Roberts, from the Walter and Eliza Hall Institute of Medical Research, University of Melbourne, Australia, gave a presentation on the topic of “Targeting BCL2 and MCL 1 in blood cancers: Practice, pitfalls, and possibilities.” This presentation was part of the session titled “Novel Targets in Hematologic Malignancies”.
Background:
Professor Roberts began his talk by explaining why BCL2 proteins are a good target in cancer treatment. The BCL2 family of proteins regulates apoptosis through both pro- and anti-apoptotic family members. The pro-apoptotic family members are typically BH3-only proteins and include BIM, PUMA, and BID. The anti-apoptotic BCL2 family members include Bcl-2 and MCL1. Ordinarily, anti-apoptotic proteins inhibit Bax/Bak and prevent the release of cytochrome-c and downstream activation of caspases thereby inhibiting apoptosis. After experiencing cellular stress, the pro-apoptotic BH3-only proteins inhibit the anti-apoptotic proteins, thereby relieving the inhibition of Bax/Bak and bringing about apoptosis.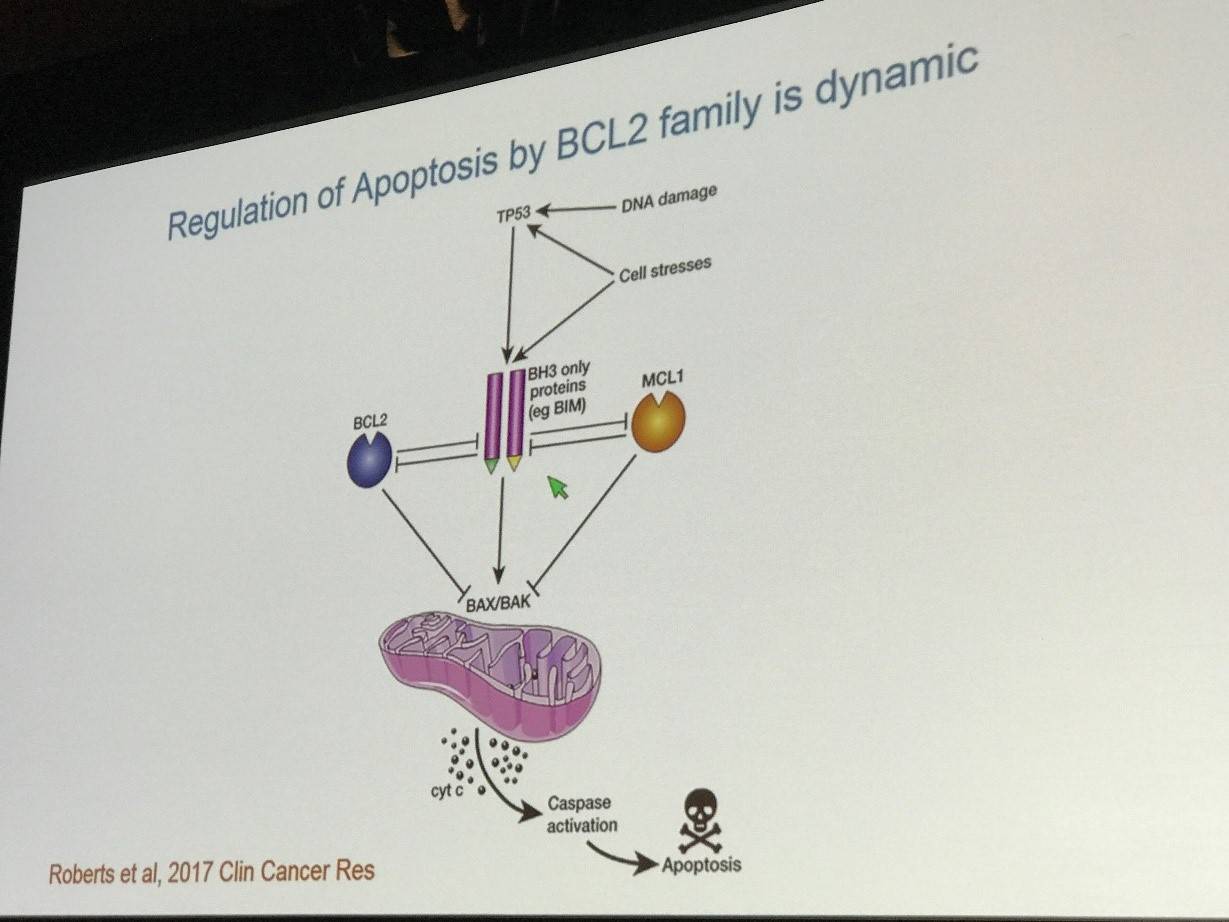
It was stated that BCL2 proteins are known to be overexpressed in blood cancers, including through a constitutive t(14;18) mutation in FL and Double-Hit Lymphoma, as well as through loss of negative regulation in CLL via miRNA 15, 16. Furthermore, it was stated that epigenetic upregulation of MCL1, resulting in overexpression, is also common in blood cancers. Together this provides a rationale for targeting BCL2 and MCL1.
BH3 mimetics act by inhibiting the extra copies of anti-apoptotic BCL2 proteins, through mimicking the binding of a BH3-only protein. This allows for the pro-apoptotic signal to be relayed to Bax/Bak to release cytochrome-c. The first such mimetic was ABT-737 and navitoclax which inhibit three anti-apoptotic proteins: Bcl-2, Bcl-Xl, and Bcl-w. Venetoclax is different from ABT-737 and navitoclax in that it selectively inhibits only Bcl-2. 
S63845 selectively inhibits MCL1. Two other small molecules were discussed during AACR 2017 which inhibit MCL1; these were AZD5991 and AMG176.
Anti-BCL2:
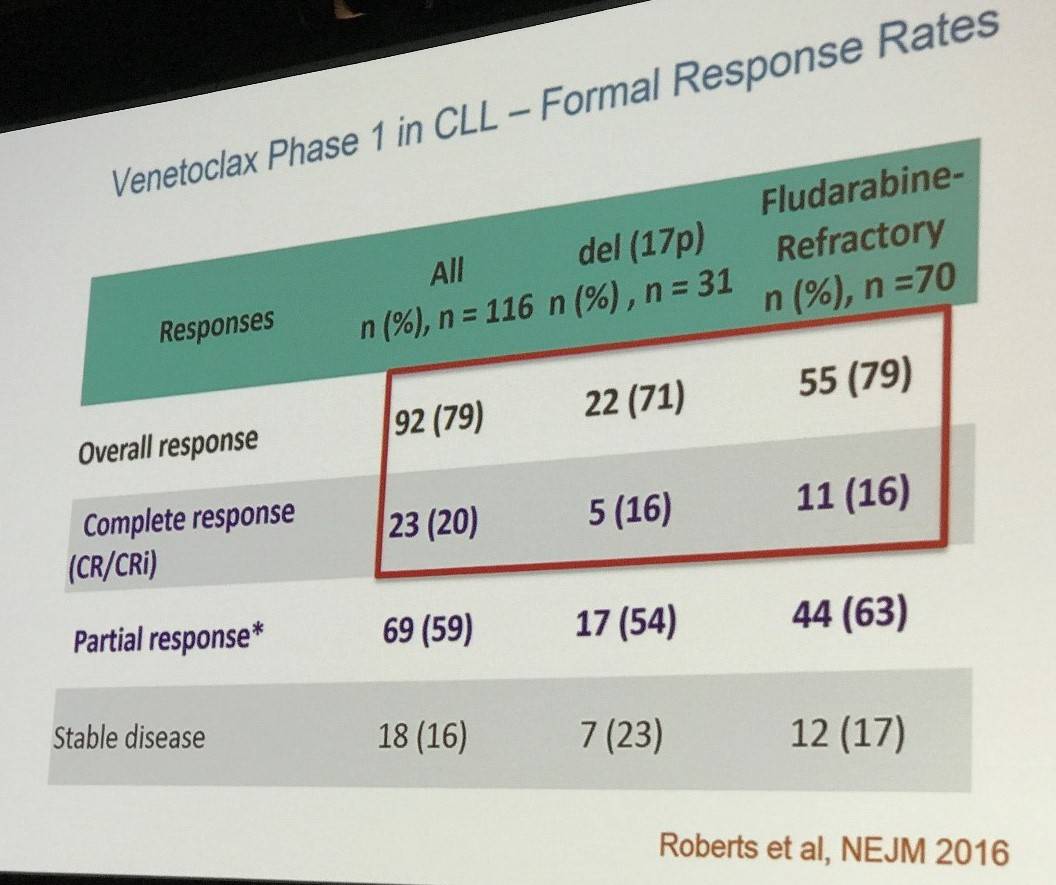
Initial in vivo trials performed with venetoclax in CLL showed positive data at 100–200mg daily dose, including a rapid improvement in CLL burden, which was replicated in in vitro studies. During a dose-escalation phase I study, it was found that 400mg daily was the optimum dose for a phase II trial, given the higher toxicities reported at higher doses. The response rates from this trial were also presented.
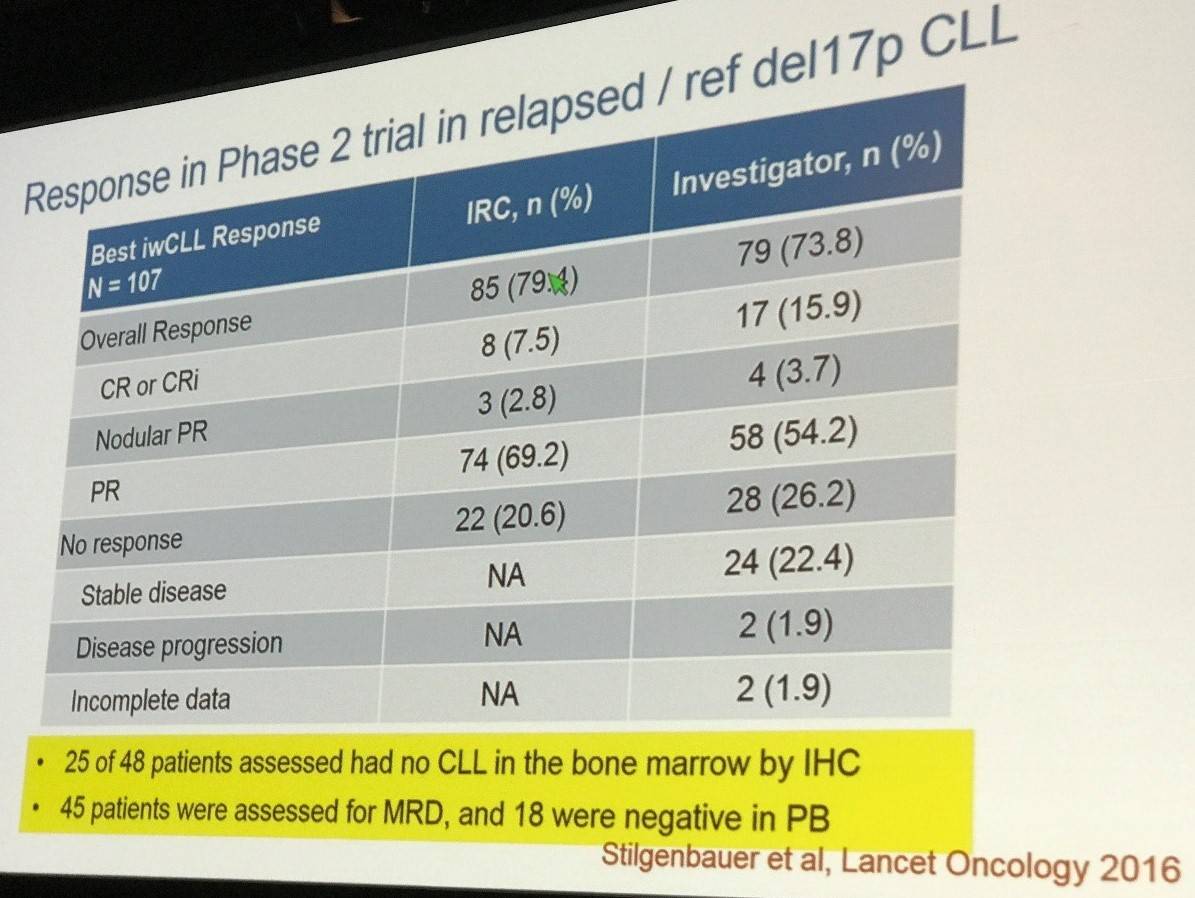
The subsequent venetoclax phase II study was also discussed this time with R/R CLL patients who had the del17p mutation. Data included a 12-month estimated PFS of 72%, and OS of 86.7%. Discontinuations and dose reductions at 400mg per day were shown to be ‘uncommon’, however, tumor lysis syndrome and febrile neutropenia presented in 3% and 6% of patients, respectively.
Subsequently, venetoclax has gone on to be trialed in other B-cell malignancies as summarized by the slide below: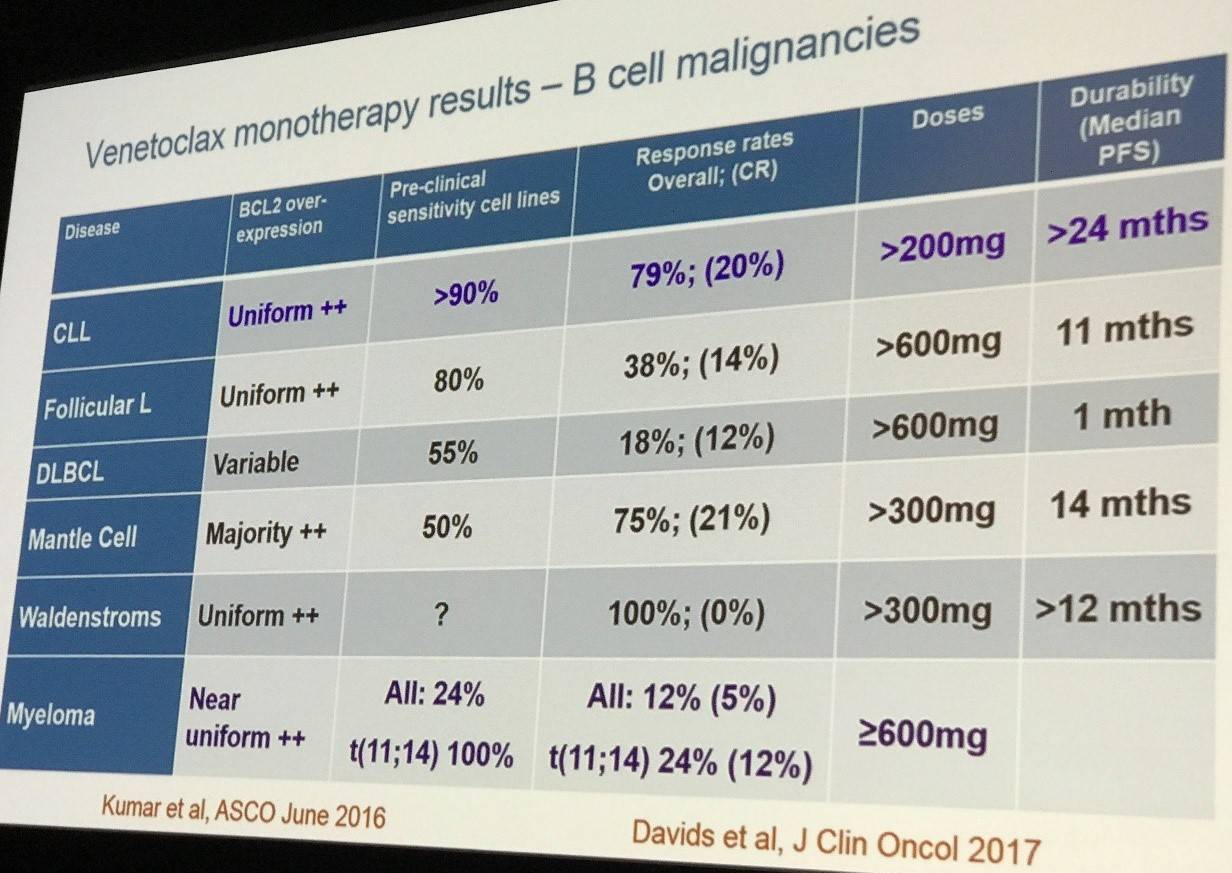

As reported in January, a phase 1b combination trial of venetoclax with rituximab has been published by John Seymour. Another study, the AIM trial in R/R MCL, is ongoing using venetoclax in combination with ibrutinib, and this data will be presented during ASCO 2017.
Anti-MCL-1:
Inhibitors of MCL-1 that were mentioned during the talk were S63845, AMG176, and AZD5991. Information presented did not include any trial data, but did include data showing that 17/25 Myeloma cell lines were sensitive to MCL-1 inhibition with S63845. Data was also shown in Acute Myeloid Leukemia (AML) cell lines, indicating that 8/25 cell lines were highly sensitive to MCL-1 inhibition with S63845. Some efficacy has also been seen in NHL.
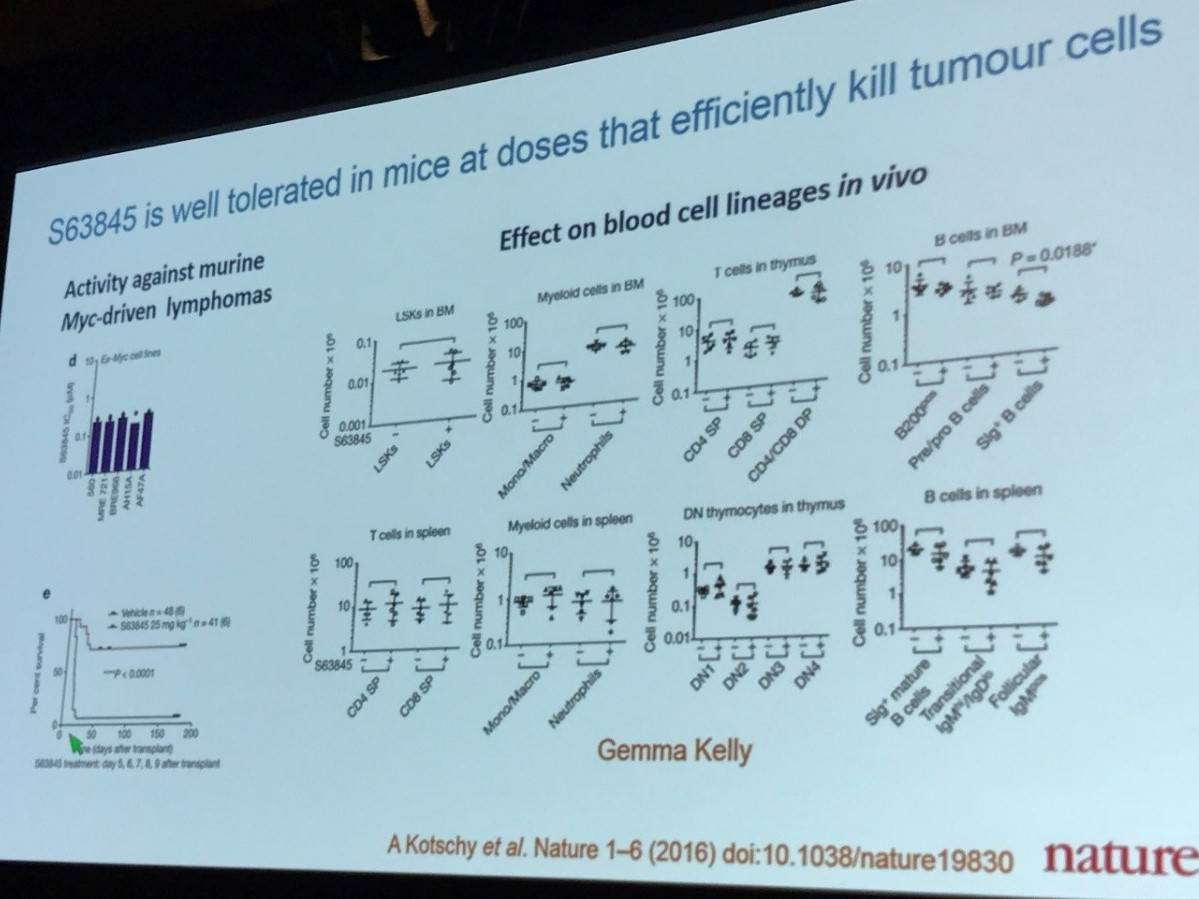

Conclusion:
In summary, Professor Roberts stated that biomarkers will be needed to find which patients’ diseases are most suited to certain inhibitors, and that both combination venetoclax trials and phase I MCL-1 trials are ongoing.
- Roberts A.W. Targeting BCL2 and MCL 1 in blood cancers: Practice, pitfalls, and possibilities [Presentation]. In: Proceedings of the 107th Annual Meeting of the American Association for Cancer Research; 2017 Apr 1-5; Washington, DC. Philadelphia (PA): AACR; 2017. Session nr [RADT13].

Understanding your specialty helps us to deliver the most relevant and engaging content.
Please spare a moment to share yours.
Please select or type your specialty
 Thank you
Thank youRelated articles
Newsletter
Subscribe to get the best content related to lymphoma & CLL delivered to your inbox








