All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a Healthcare Professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The Lymphoma Hub uses cookies on this website. They help us give you the best online experience. By continuing to use our website without changing your cookie settings, you agree to our use of cookies in accordance with our updated Cookie Policy
Introducing

Now you can personalise
your Lymphoma Hub experience!
Bookmark content to read later
Select your specific areas of interest
View content recommended for you
Find out moreThe Lymphoma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Lymphoma Hub cannot guarantee the accuracy of translated content. The Lymphoma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
AACR 2017 | The MYC oncogene is a master regulator of immune response: MYC inactivation presents a potential curative option for Lymphoma
Bookmark this article
This year’s American Association for Cancer Research (AACR) annual meeting took place on 1–5 April in Washington, DC, USA. The program committee Chair was Kornelia Polyak, MD, PhD, from the Dana-Farber Cancer Institute, Boston, Massachusetts.
On Sunday 2nd April, during the “RAOS02 - Immunotherapy of Lymphoid Malignancies” session, Dean W. Felsher, MD, PhD, from the Stanford University School of Medicine, gave a talk titled: “The Myc Oncogene is a Master Regulator of the Immune Response.”
Dr Felsher began the talk by highlighting that oncogene dependency can be a cancer’s “Achilles heel”; knocking out the oncogene can cause cancer cells to differentiate (displaying a normal phenotype) or to undergo apoptosis.
MYC is a transcription factor with multiple functions and plays a role in a wide range of cellular pathways; because of this, MYC contributes to multiple cancer traits:
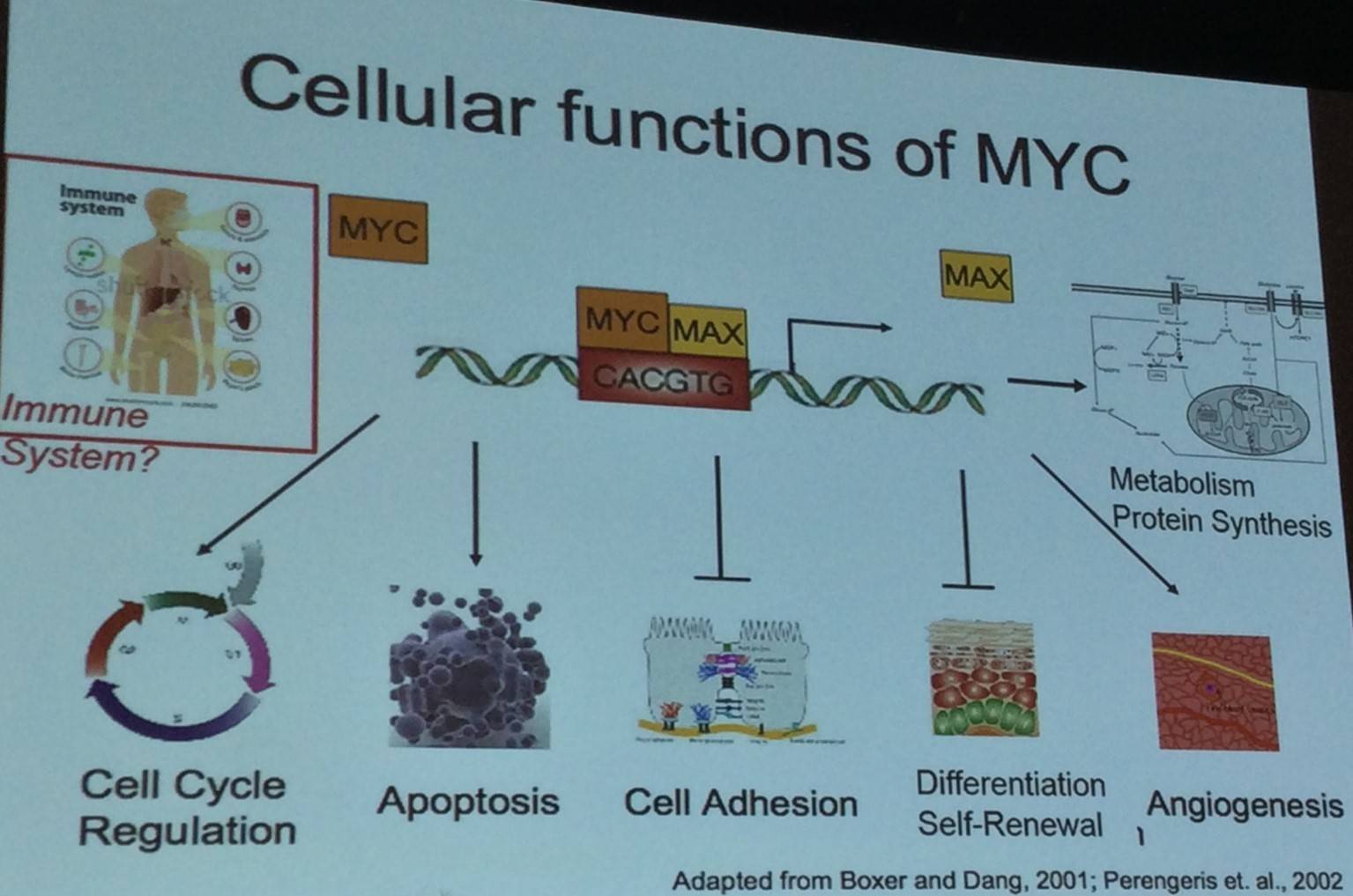
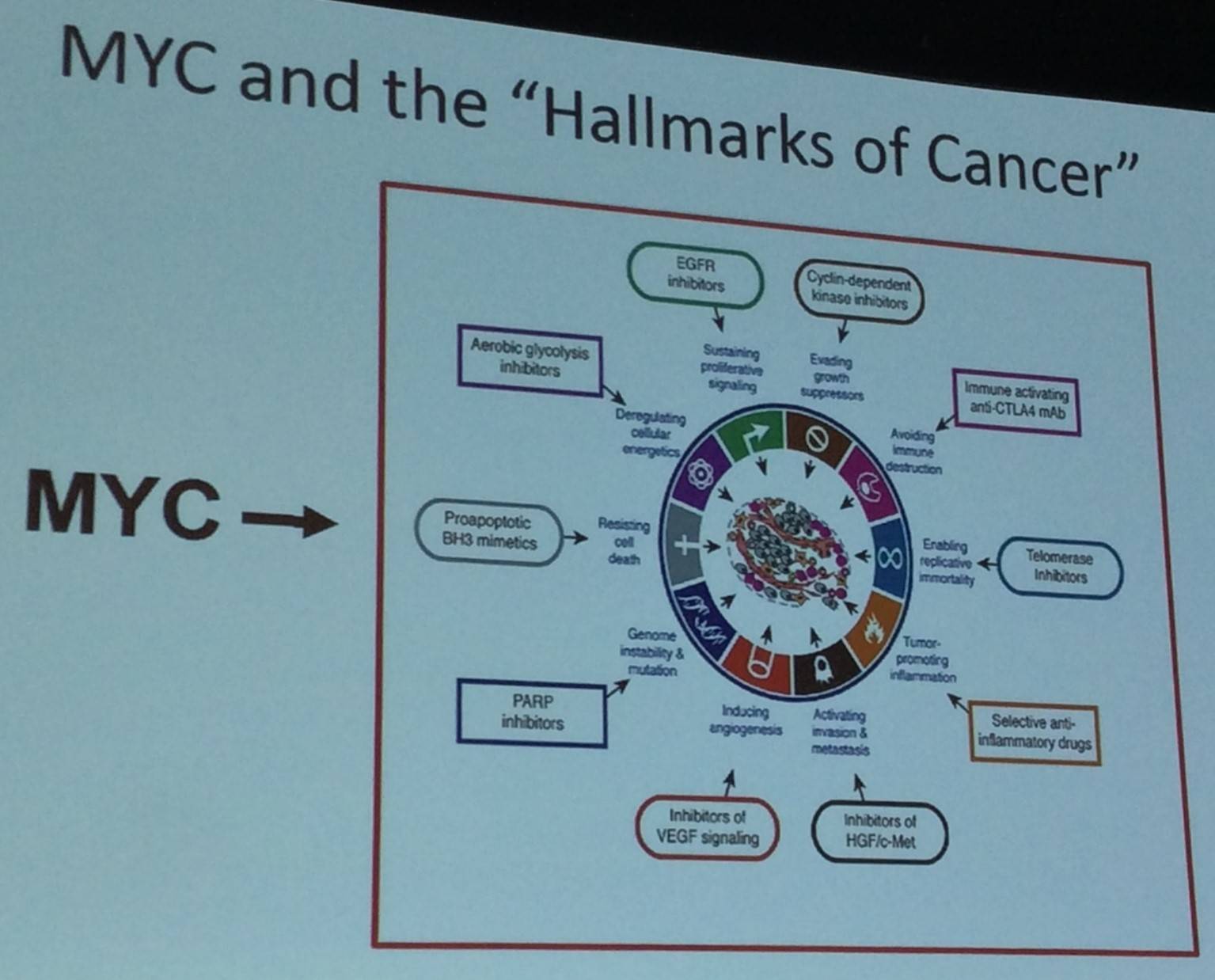
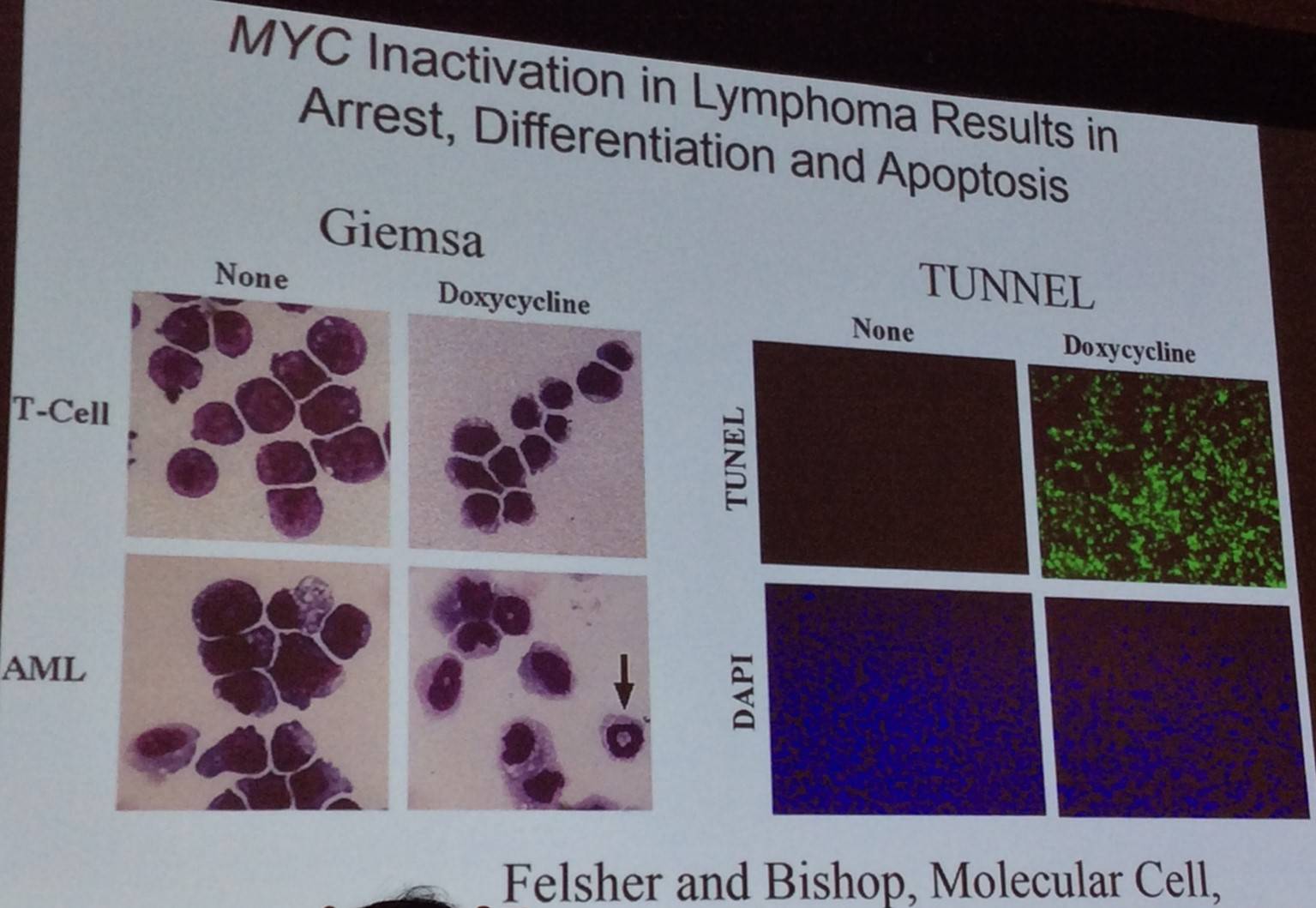
Dr Felsher went on to explain that in a tet inducible mouse model, enabling MYC to be turned on and off, it was found that MYC induced lymphomagenesis can be reversed. Moreover, switching MYC off in lymphoma resulted in cell cycle arrest, differentiation, and apoptosis:
The talk then discussed the role of MYC in cellular senescence: suppression of MYC results in cellular senescence in a variety of tumor types, including lymphoma.
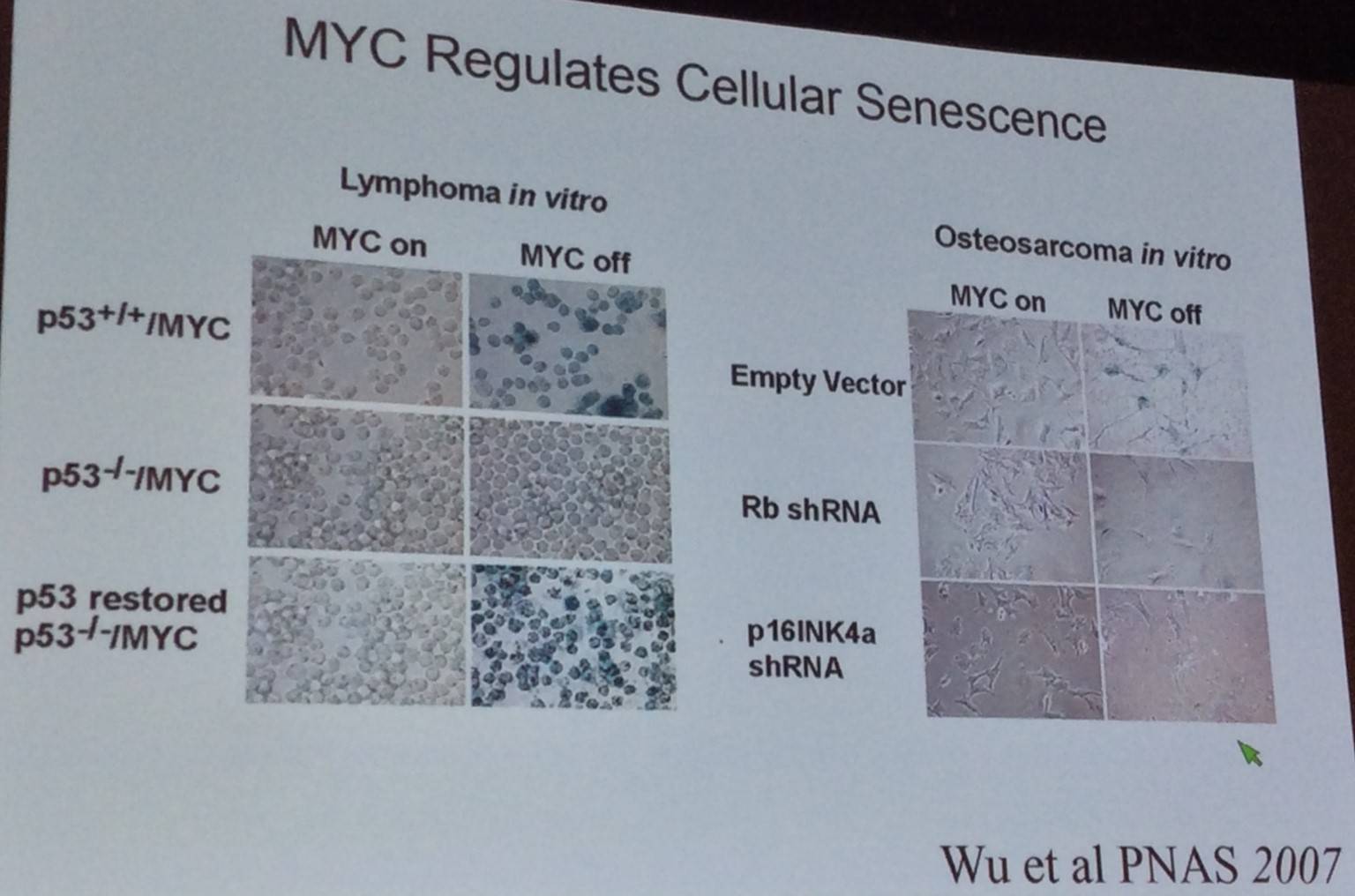
Following this, Dr Felsher outlined findings published by Wu et al. in the Proceedings of the National Academy of Sciences of the United States of America in 2007: programs of cellular senescence stay latently functional, even in established tumors, and these programs can be activated again, providing a crucial mechanism of oncogene addiction associated with inactivation of MYC.
An overview was then given of how inactivation of MYC results in regression of tumors:
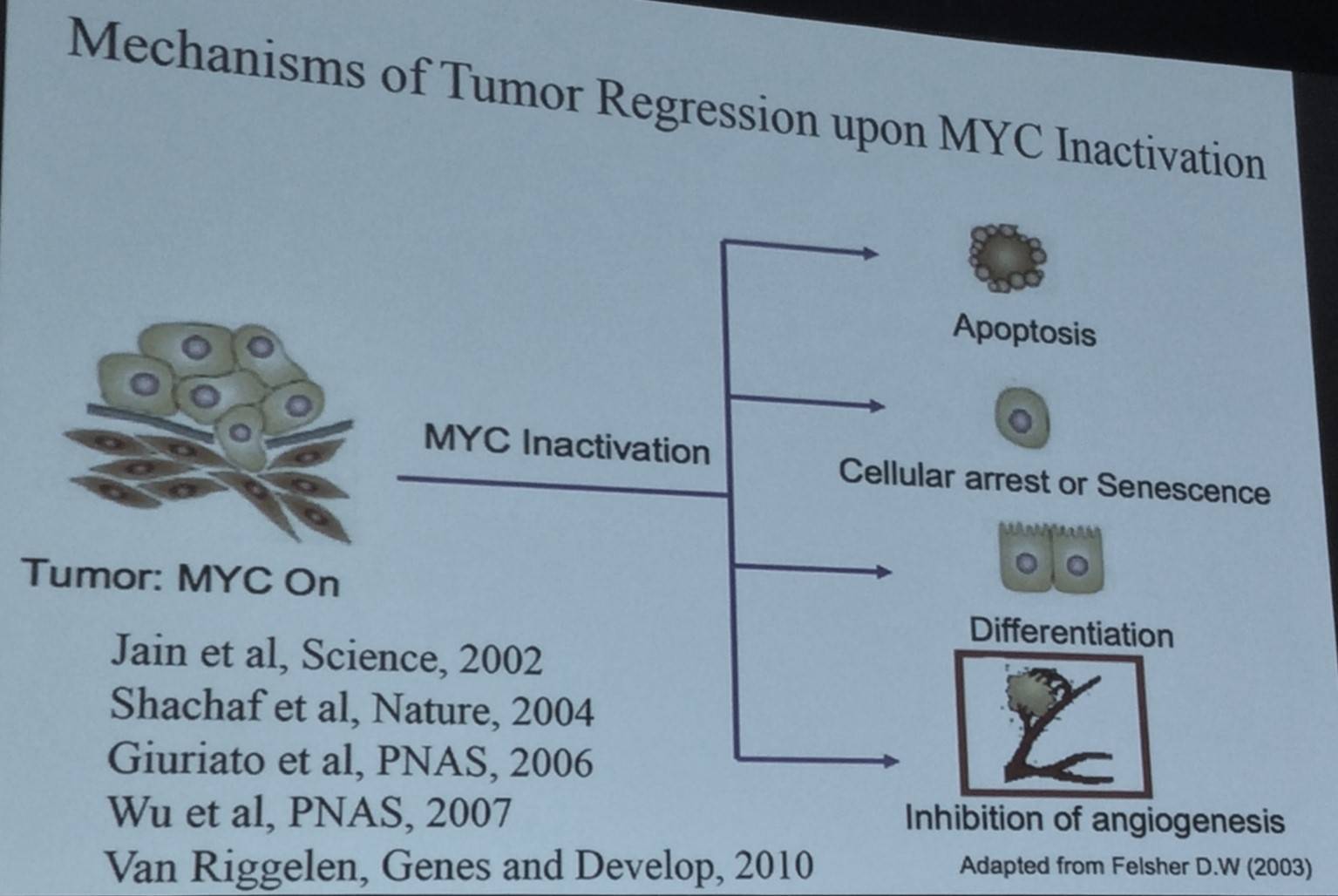
The focus of the talk then turned to whether MYC regulates the immune system and if this influences stemness versus senescence?
An active immune system is crucial for continual regression of tumors, for stimulating oncogene addiction, and CD4+ T-cells specifically are needed to induce cellular senescence after inactivation of MYC (Rakhra et al. Cancer Cell, 2010).
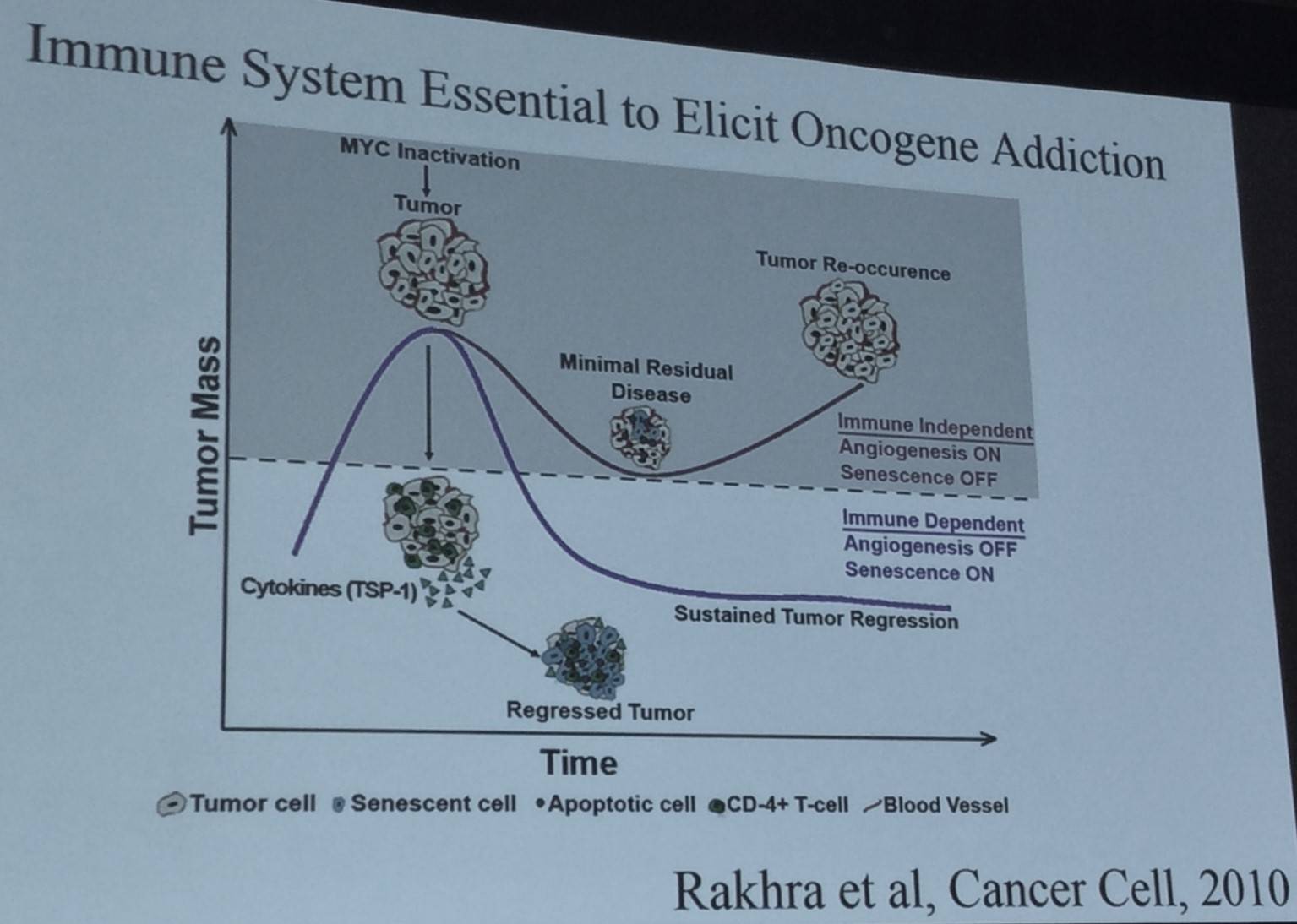
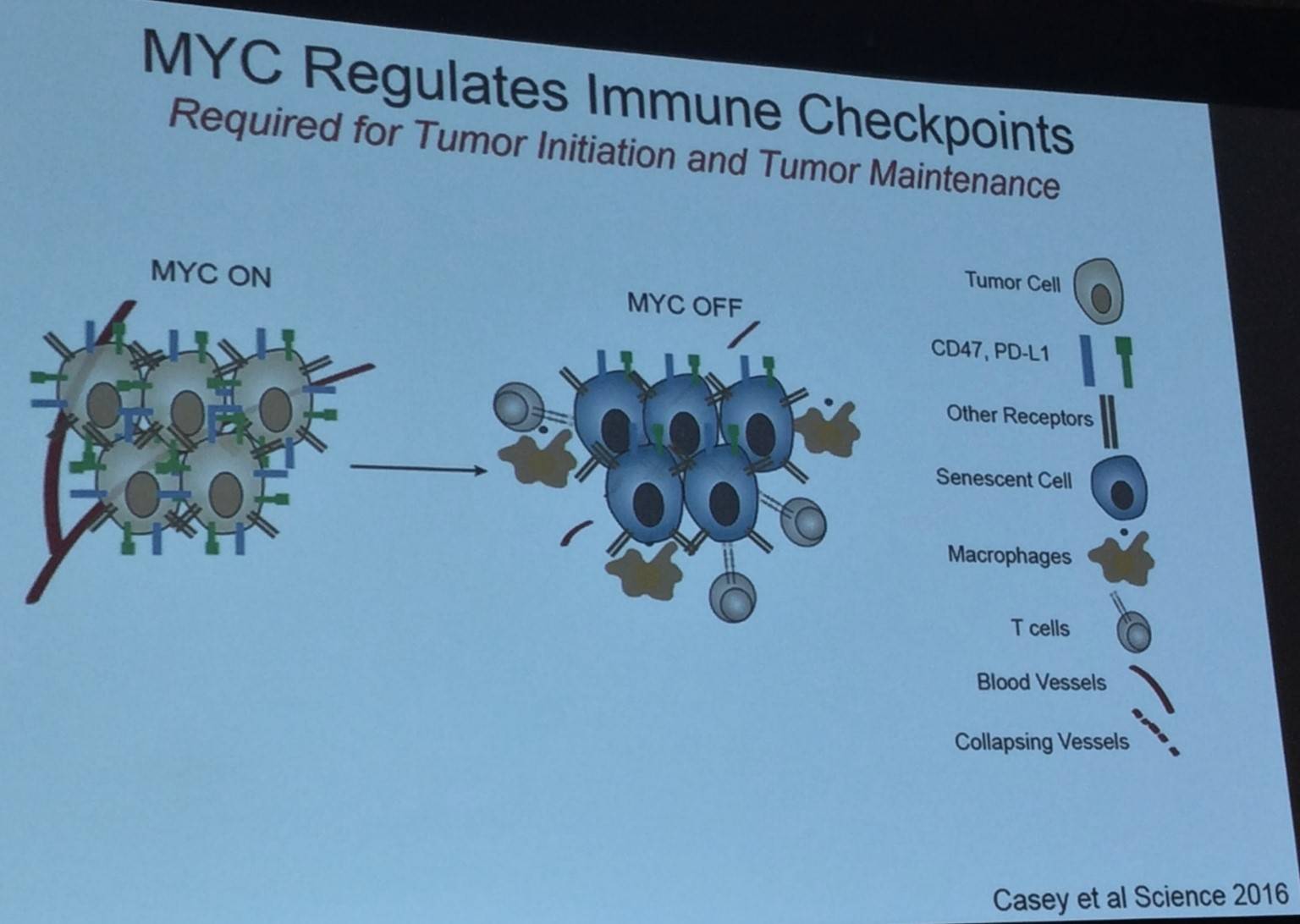
Next, Dr Felsher discussed recent findings published by Casey et al. in Science in 2016:
- MYC regulates expression of two immune checkpoint proteins on the surface of tumor cells: CD47 (innate immune regulator) and PD-L1 (adaptive immune checkpoint)
- MYC binds directly to Cd47 and Pd-l1 genes promoters
- Silencing MYC in mouse tumors and human tumor cells decreased levels of CD47 and PD-L1 mRNA and protein, and enhanced the antitumor immune response
However, inactivating MYC in tumors with enforced expression of CD47 or PD-L1 resulted in repression of the immune response and continual tumor growth
Dr Felsher then presented an overview slide of how oncogenes and the immune response interact:
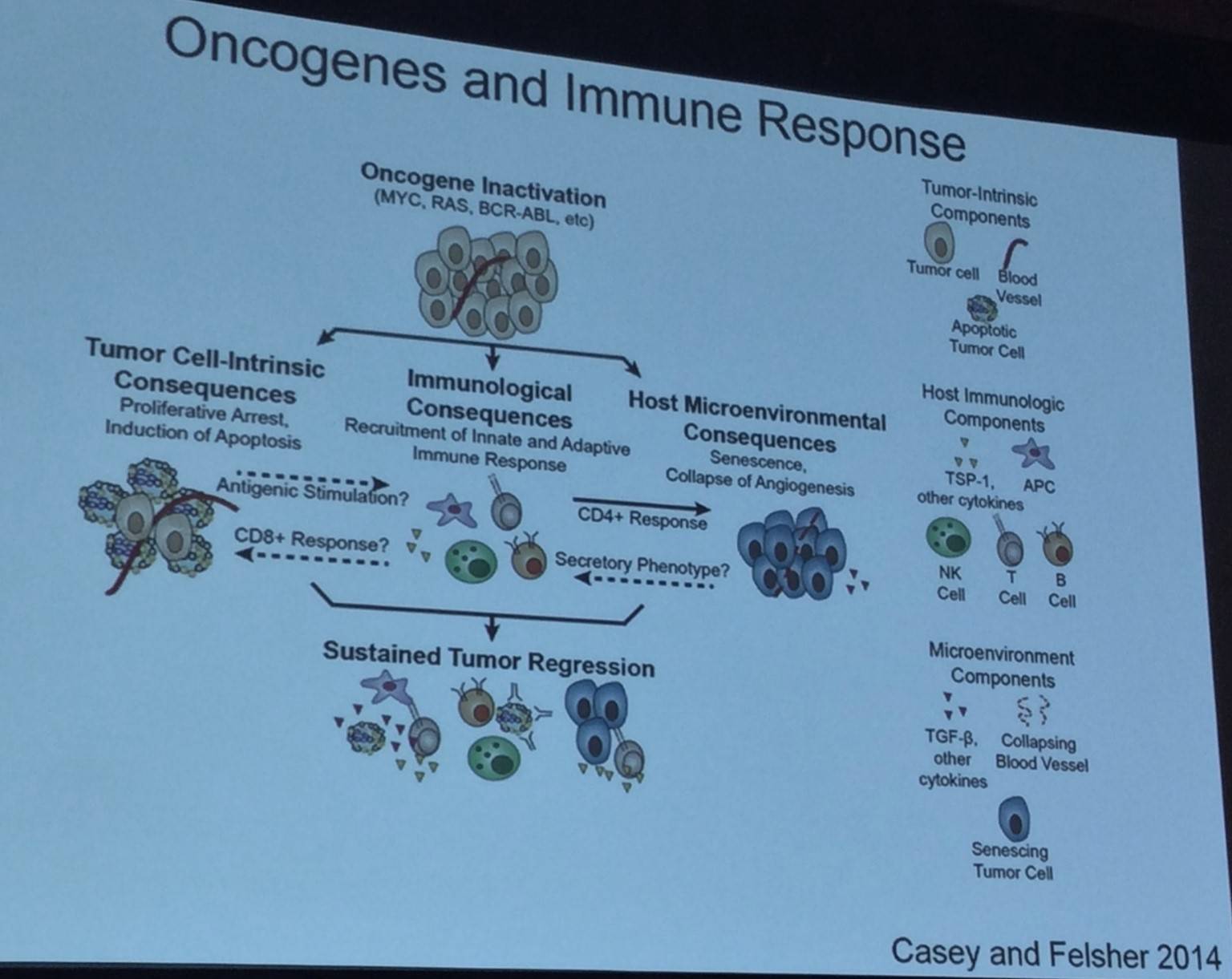
The talk was concluded by stating that cancers, including Lymphomas such as Burkitt Lymphoma, Mantle Cell Lymphoma, and Double Hit Lymphoma, may potentially be cured by inactivating the MYC oncogene. MYC inactivation reinstates the immune response against cancer. Dr Felsher also suggested that nanoscale proteomics (NIA) provide a potential strategy of identifying oncogene activation in patients, informing therapeutic choice.
- Felsher D.W. The Myc Oncogene is a Master Regulator of the Immune Response. 2017 Apr 2. AACR Annual Meeting 2017. Washington, DC, USA.

Understanding your specialty helps us to deliver the most relevant and engaging content.
Please spare a moment to share yours.
Please select or type your specialty
 Thank you
Thank youRelated articles
Newsletter
Subscribe to get the best content related to lymphoma & CLL delivered to your inbox








