All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a Healthcare Professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The Lymphoma Hub uses cookies on this website. They help us give you the best online experience. By continuing to use our website without changing your cookie settings, you agree to our use of cookies in accordance with our updated Cookie Policy
Introducing

Now you can personalise
your Lymphoma Hub experience!
Bookmark content to read later
Select your specific areas of interest
View content recommended for you
Find out moreThe Lymphoma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Lymphoma Hub cannot guarantee the accuracy of translated content. The Lymphoma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
iwCLL 2017 | Additional signaling pathways in CLL and their interactions with the BCR
Bookmark this article
On 14th May 2017, the “Role of the B-Cell Receptor and Other Signaling Pathways in CLL” took place at iwCLL, and was co-chaired by Nicholas Chiorazzi (The Feinstein Institute for Medical Research, Manhasset, NY, USA) and Kostas Stamatopoulos (Center for Research and Technology Hellas, Thessaloniki, Greece).
During this session, “Additional Signaling Pathways in CLL and Their Interactions With the BCR” was presented by Dimitar Efremov from the International Centre for Genetic Engineering and Biotechnology, Trieste, Italy.
The presentation began by outlining the evidence for a role of BCR signaling in the development and evolution of CLL (Iacovelli et al. 2015; Herishanu et al. 2011):
- Selection of stereotyped BCRs
- Correlation of clinical course with BCR-related features
- Clinical activity of BCR signaling inhibitors
- Selection of cell-autonomous BCRs and BCRs reactive with low-affinity autoantigens during leukemia development in Eµ transgenic mice
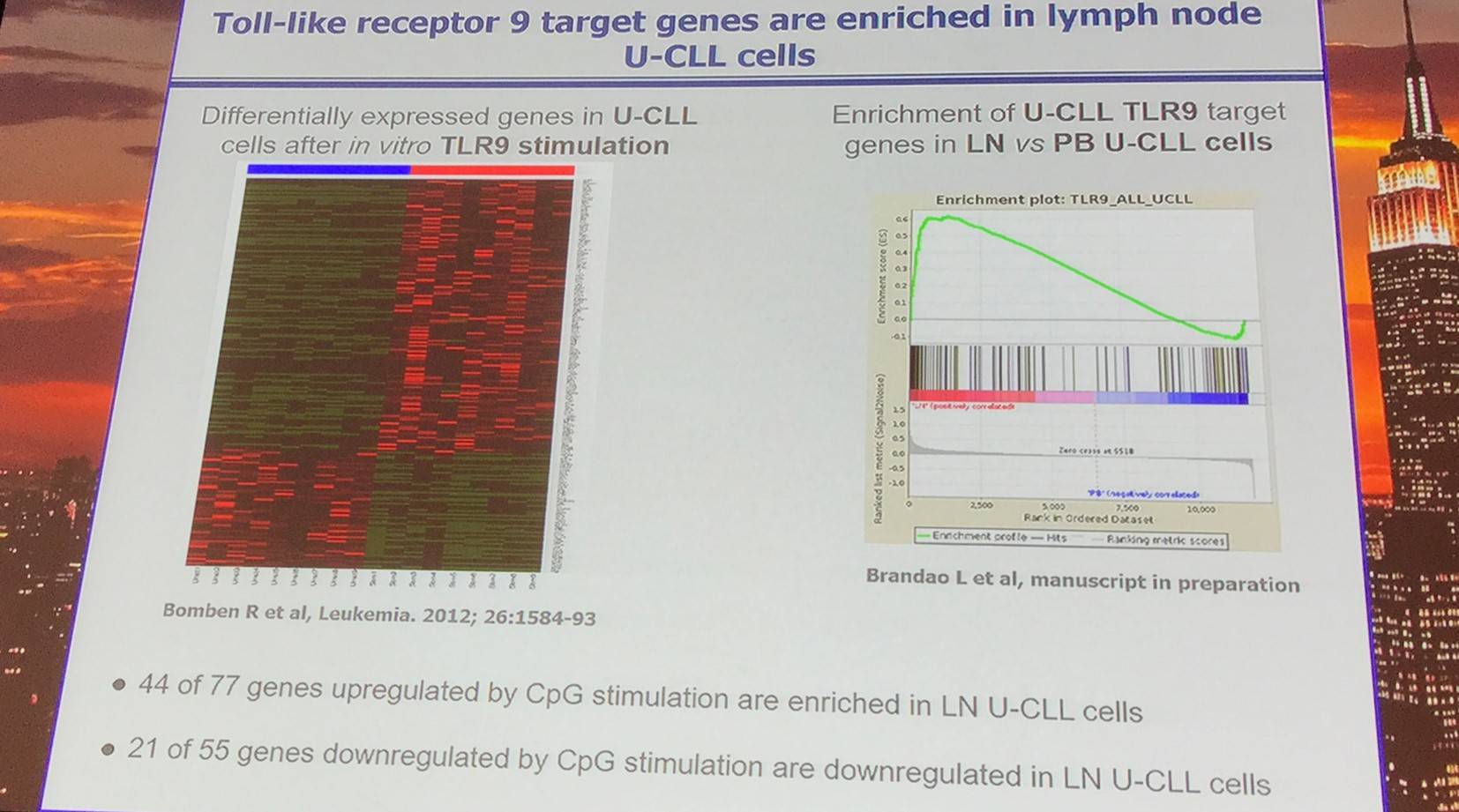
- Genes are differentially expressed:
- Between lymph node and blood CLL cells
- In CLL cells after in vitro BCR stimulation
- BCR-regulated genes are enriched in lymph node CLL cells
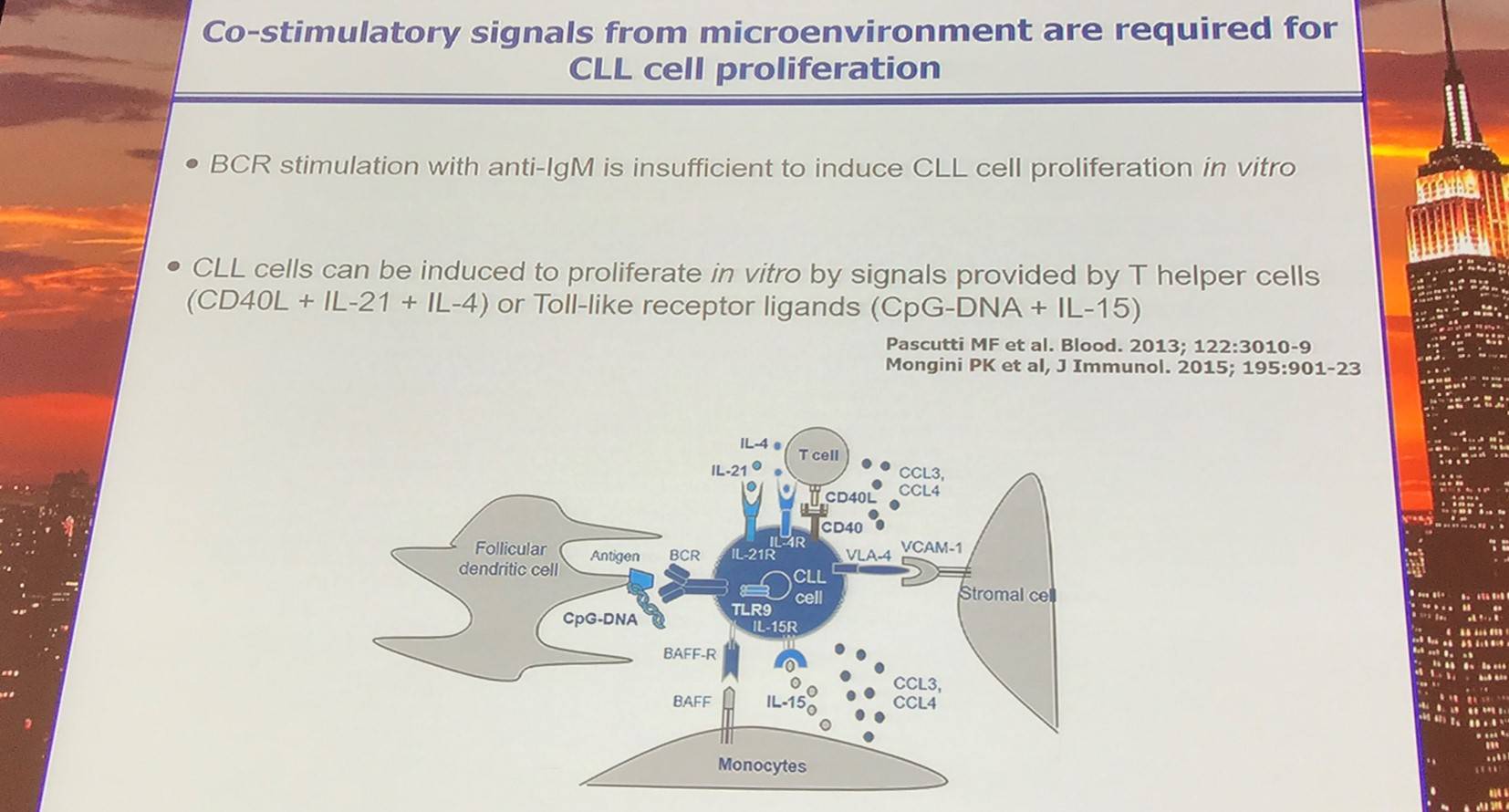
CLL cells interact with T-cells and macrophages in proliferating centers in lymph nodes. Moreover, the interaction between activated T-cells and lymph node CLL cells has been reported in vivo and results in enrichment of IL-21 and IL-4 target genes in lymph node CLL cells.
BCR regulates proliferation of CLL cells via numerous mechanisms:
- Internalization of antigen and DNA-containing immune complexes: T-cell and TLR9 activation
- Inside-out integrin activation: tissue adhesion and retention (Buchner et al. 2010)
Dimitar Efremov asked is BCR signals are required for CLL proliferation? In patients and Eµ-TCL1 transgenic mice, BTK and SYK inhibitors rapidly inhibit proliferation of CLL cells. Moreover, stimulation of surface IgM induces expression of positive cell cycle regulators (MYC, CCND2) in a subset of CLL samples (Krysov et al. 2012). In patients and mouse models, BCR responsiveness positively correlates with disease severity and, in Eµ-TCL1 transgenic mice, enhanced BCR signaling correlates with enhanced proliferation of CLL cells.

There are a number of potential mechanisms to explain reduced surface IgM expression and BCR signaling capacity of CLL cells in the peripheral blood (Mockridge et al. 2007; Dühren-von Minden et al. 2012):
- Interactions with external autoantigens
- Cell-autonomous BCR-BCR interactions
- Interactions with soluble or FCµR-bound serum IgM
It has been found that serum IgM inhibits BCR signaling and that FCµR triggering alone is also sufficient to inhibit BCR signaling.
Additionally, IL-4 has been reported to upregulate expression of surface IgM and downregulates expression of FCµR on CLL cells. Also, FCµR expression is lower on lymph node CLL cells vs. peripheral blood CLL cells.
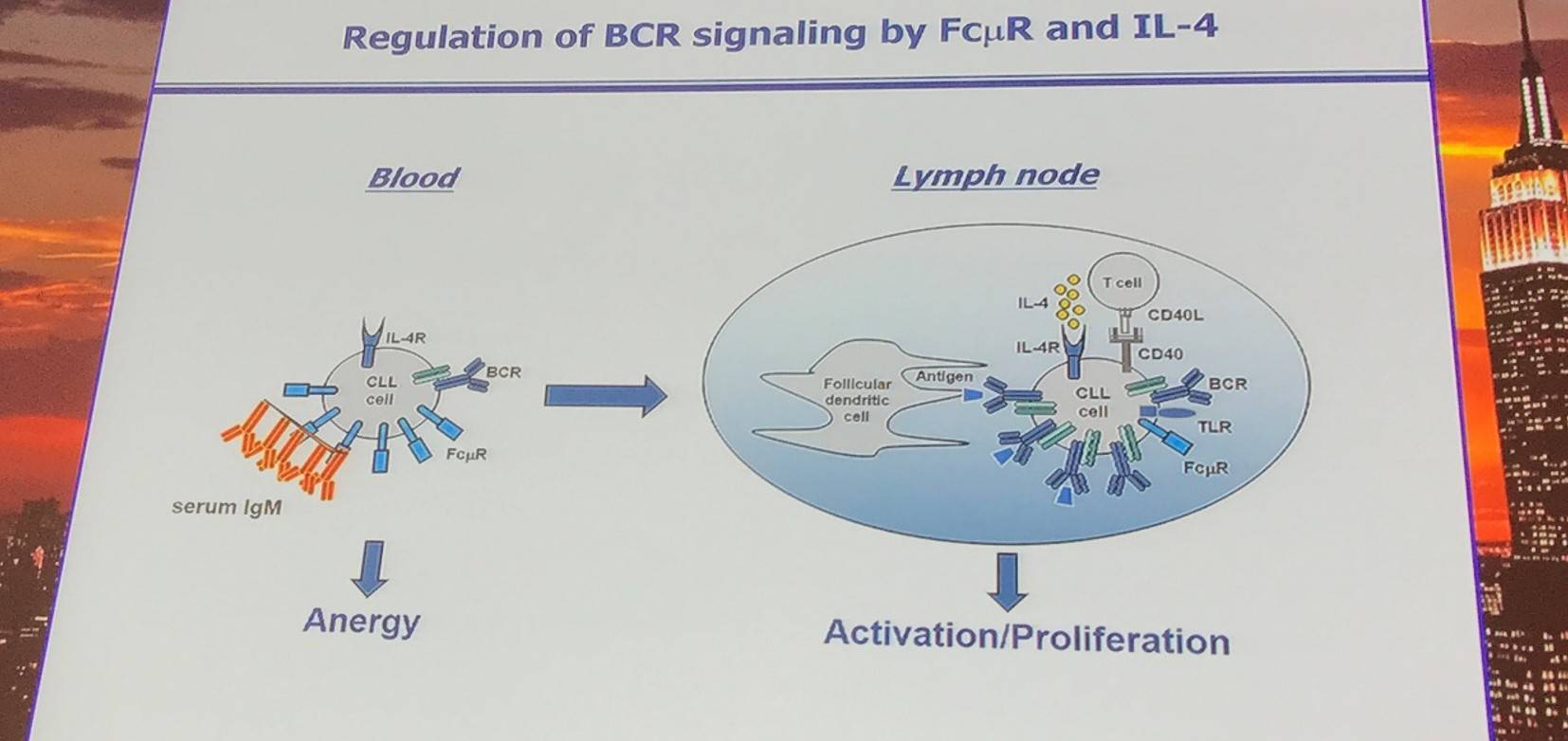
Moreover, serum IgM levels correlate with Treatment-Free Survival in U-CLL and inhibits expansion of human CLL cells xenografted in immunodeficient NSG mice.
Low FCµR levels are associated with a short Treatment-Free Survival and accelerates leukemia development in Eµ-TCL1 transgenic mice.
Dimitar Efremov concluded the talk with a concise summary slide:

- Efremov D. Additional Signaling Pathways in CLL and Their Interactions With the BCR. XVII International Workshop on Chronic Lymphocytic Leukemia; 2017 May 12–15; New York, USA.

Understanding your specialty helps us to deliver the most relevant and engaging content.
Please spare a moment to share yours.
Please select or type your specialty
 Thank you
Thank youRelated articles
Newsletter
Subscribe to get the best content related to lymphoma & CLL delivered to your inbox








