All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a Healthcare Professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The Lymphoma Hub uses cookies on this website. They help us give you the best online experience. By continuing to use our website without changing your cookie settings, you agree to our use of cookies in accordance with our updated Cookie Policy
Introducing

Now you can personalise
your Lymphoma Hub experience!
Bookmark content to read later
Select your specific areas of interest
View content recommended for you
Find out moreThe Lymphoma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Lymphoma Hub cannot guarantee the accuracy of translated content. The Lymphoma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
ASH 2016 | Educational Session: Response Adapted Frontline Therapy in Hodgkin Lymphoma: Are We There Yet?
Bookmark this article
On Saturday 3rd December, during the 58th Annual Meeting & Exposition of the American Society of Hematology (ASH), in San Diego, CA, an educational session took place between 9.30am–11.00am, titled “Hodgkin Lymphoma: Treatment Today and the Challenge for Tomorrow.” This session was Chaired by Andrew Lister, MD, of Barts Cancer Institute, London, UK, and focused on how the outcome of patients with HL in 2016 can be improved, beyond that achieved in the last 50 years, with combination chemotherapy and extended-field megavoltage radiotherapy.
“Response Adapted Frontline Therapy in Hodgkin Lymphoma: Are We There Yet?” was the first talk during this session, and was presented by Peter M. Johnson, MA, MD, of the University of Southampton, UK, and discussed results of major trials evaluating the role of functional imaging with FG PET to inform escalation or de-escalation of treatment according to the result.
Dr Johnson began by explaining the current dilemma of treating patients with HL; the probability of cure has risen however, in early and advanced stage disease, the risk of death from disease is exceeded by the risk of death from other causes, particularly the toxic effects of therapy. Survivors are at risk of a range of long term toxicities, the most serious include:
- Epithelial malignancies induced by radiotherapy
- Myelodysplasia and Acute Leukemia from extensive alkylating agents
- Accelerated coronary and valvular heart disease induced by mediastinal radiotherapy and anthracyclines
- Infertility caused by high doses of alkylating agents
- Pulmonary fibrosis caused by bleomycin
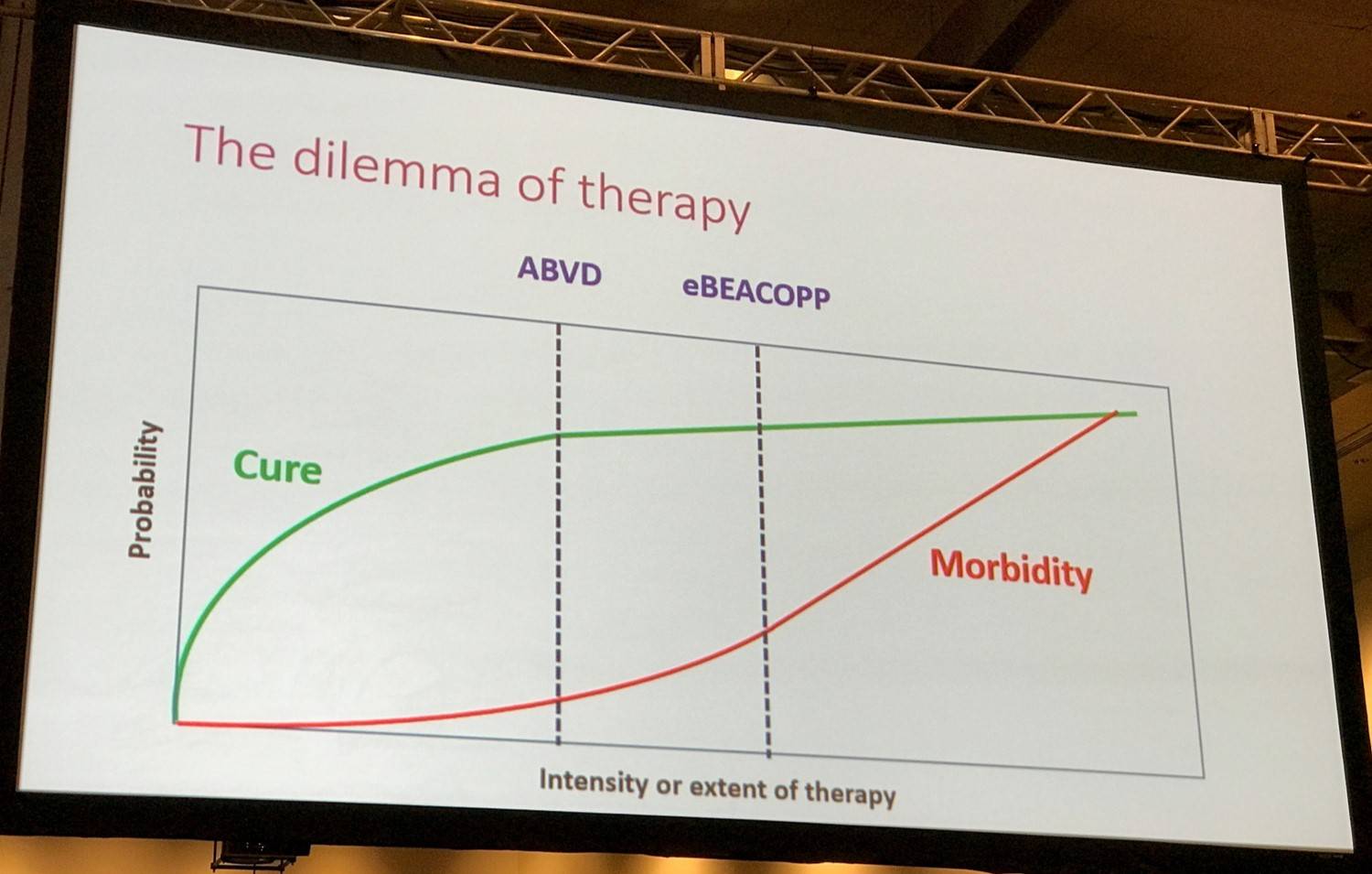
Overall, it seems that in each risk group (early stage favorable, early stage unfavorable, and advanced stage) there is a minimum treatment threshold:
- Below this level, rates of durable remission begin to decrease
- Treatment intensification higher than this threshold produces toxicities which off-set the effect of better lymphoma control
A potential solution to this problem is to use response-adapted therapy, where the effect of treatment is measured early on to identify patients in which the therapy is effective and those in which it is failing and requires modification. This is assessed by FDG-PET, based upon the preferential uptake of labeled FDG by actively metabolizing tissues such as sites of involvement by active HL, but also other tissues such as the myocardium, brain, kidney, liver, regenerating bone marrow, and brown fat. The last can cause complications when performing FDG-PET in teenage and young adult HL patients.
Dr Johnson then moved on to discuss the results of numerous groups who have conducted prospective randomized trials testing the theory of reducing or de-escalating treatment after obtaining a negative PET result.
Randomized trials to test the role of interim FDG-PET in early stage disease
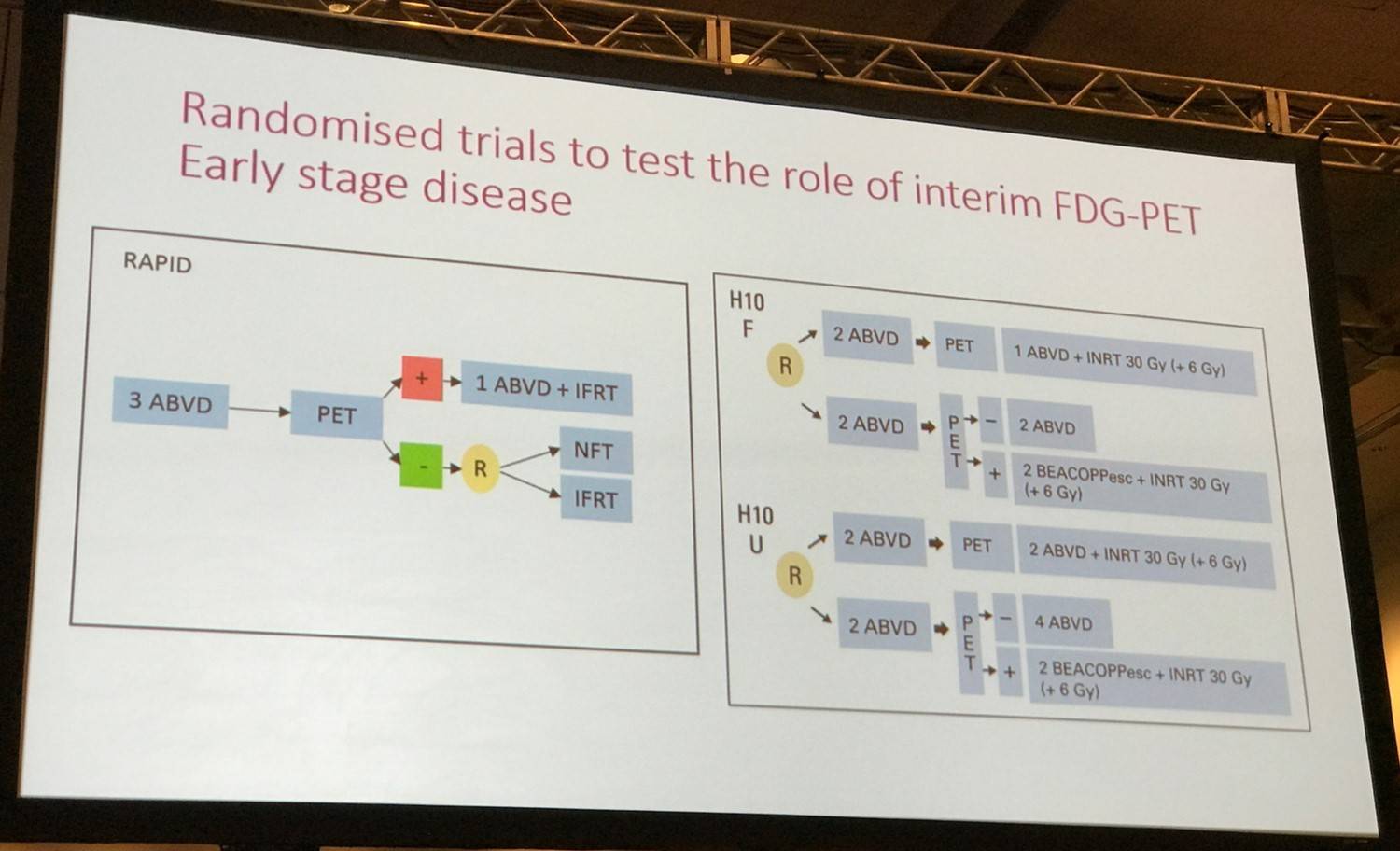
The NCRI RAPID study and the EORTC HD10 study were discussed:
- In both of these trials, there was a small decrease of PFS with reduction of treatment
- RAPID: PFS at 3 years was 94.6% in the group that received radiation therapy and 90.8% in the group that did not receive radiation therapy
- HD10: early interim analysis found that PET-guided therapy did not show equivalence to standard treatment, although in both risk strata the results were good; 1-year PFS rates were 100% and 94.9% in the favorable-risk group and 97.3% and 94.7% in the unfavorable risk group
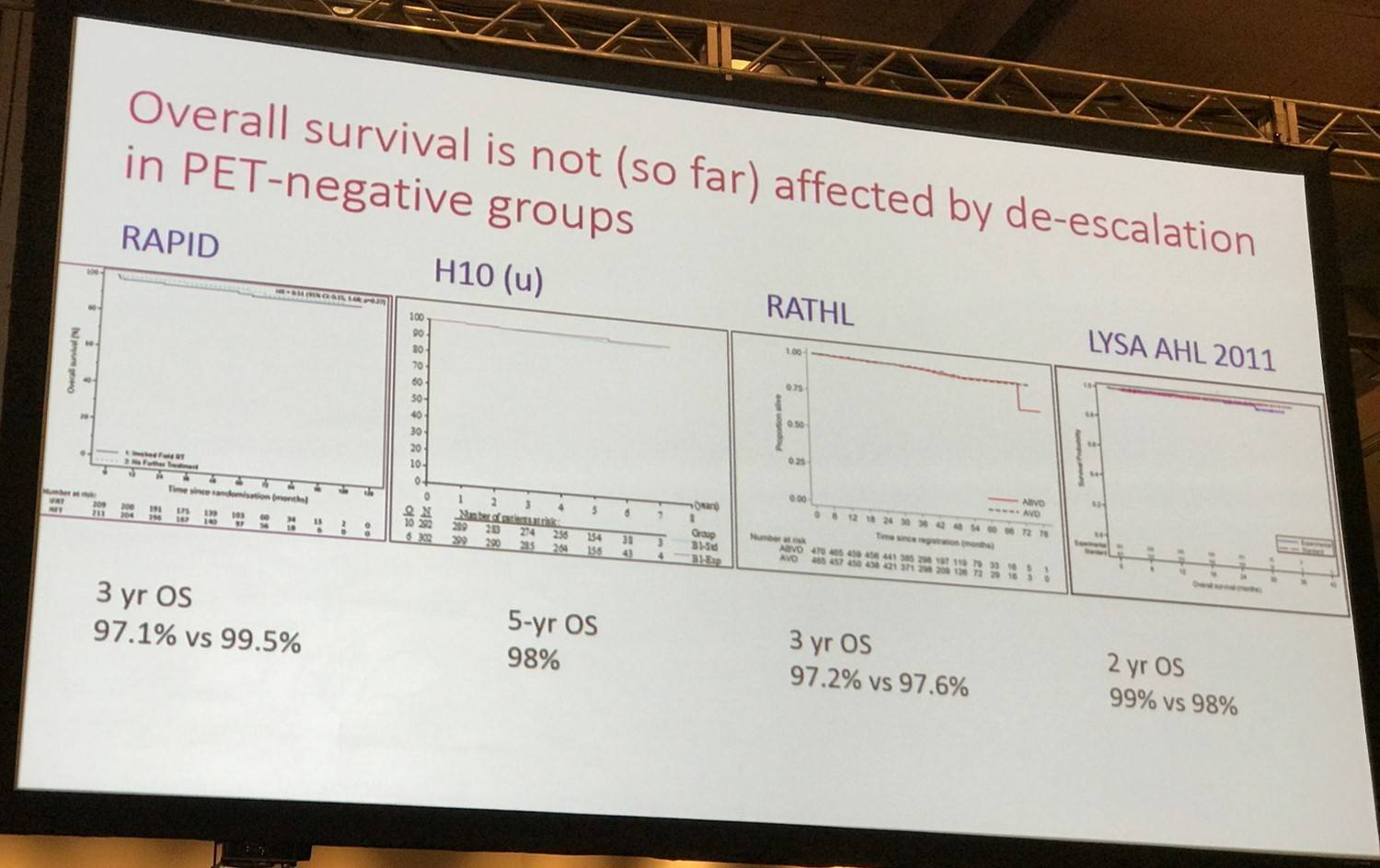
- In both RAPID and HD10, OS was shown not to be compromised by the removal of radiotherapy, although the HD10 study had a relatively short follow-up
- Absolute patient deaths were low, the RAPID study reported 12 deaths out of 420 PET-negative patients at a median follow-up of 5 years and only one death was attributed to HL
The conclusion from these trials is that removal of consolidation radiotherapy after a negative PET result in early stage HL results in a small increase in the risk of recurrence but does not worsen OS. This allows patients and their caregivers to make individual decisions based upon the relative risks from consolidation radiotherapy or the possible need for second-line treatment.
Randomized trials to test the role of interim FDG-PET in advanced stage disease
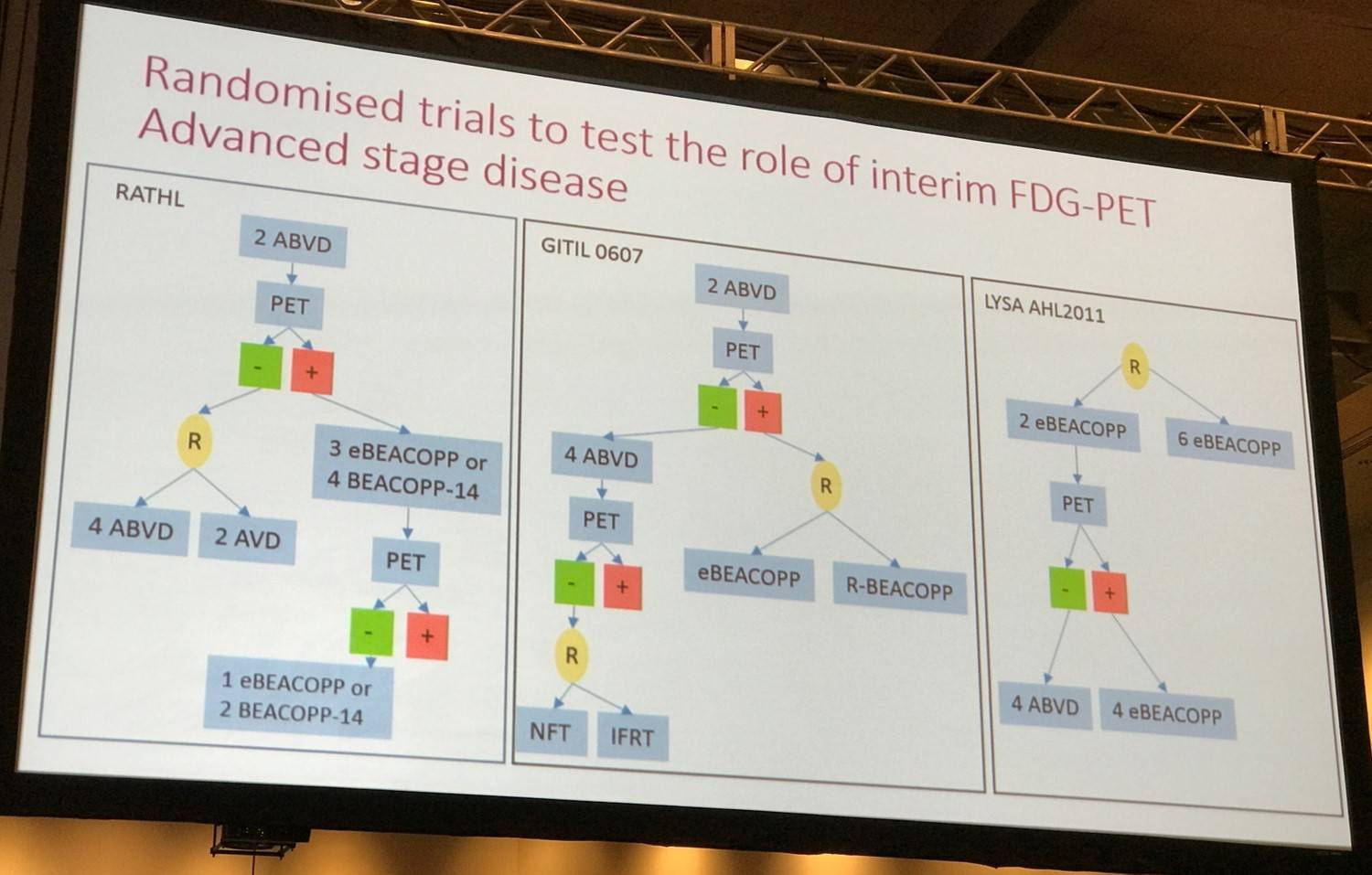
The RATHL and LYSA AHL2011 studies were discussed.
- RATHL:
- 3-year PFS was 9.6% for those with stage IV disease but 90.0% for those with stage II disease
- Presence of bulk not associate with a higher rate of recurrence after a negative interim PET scan; 3-year PFS of 87.6%
- Removal of bleomycin after a negative interim PET scan does not significantly increase the risk of recurrence in any sub-group, including advanced stage disease, high prognostic sore, or bulky disease suggesting that although the test is imperfect, it may still be used to modulate therapy
- Removal of bleomycin from the last 4 cycles of treatment resulted in fewer severe respiratory adverse events and better preservation of lung diffusion capacity
- AHL2011:
- PET-negative patients showed a 2-year PFS of 94% for the standard treatment arm and 92% for the experimental arm
Overall, this indicates that a PET-negative result is most reliable in patients with less extensive disease at presentation and in those treated with intensive chemotherapy. Across the randomized studies currently reported, the results of de-escalating therapy after a negative interim PET scan seems to be favorable. In early stage disease, the small increase in recurrence rate following the removal of radiotherapy after treatment with ABVD does not appear to reduce OS. Moreover, in advanced stage disease, neither removing bleomycin after treatment with ABVD nor reducing treatment from BEACOPPescalated to ABVD appears to increase recurrence risk, although there has been relatively limited follow-up.
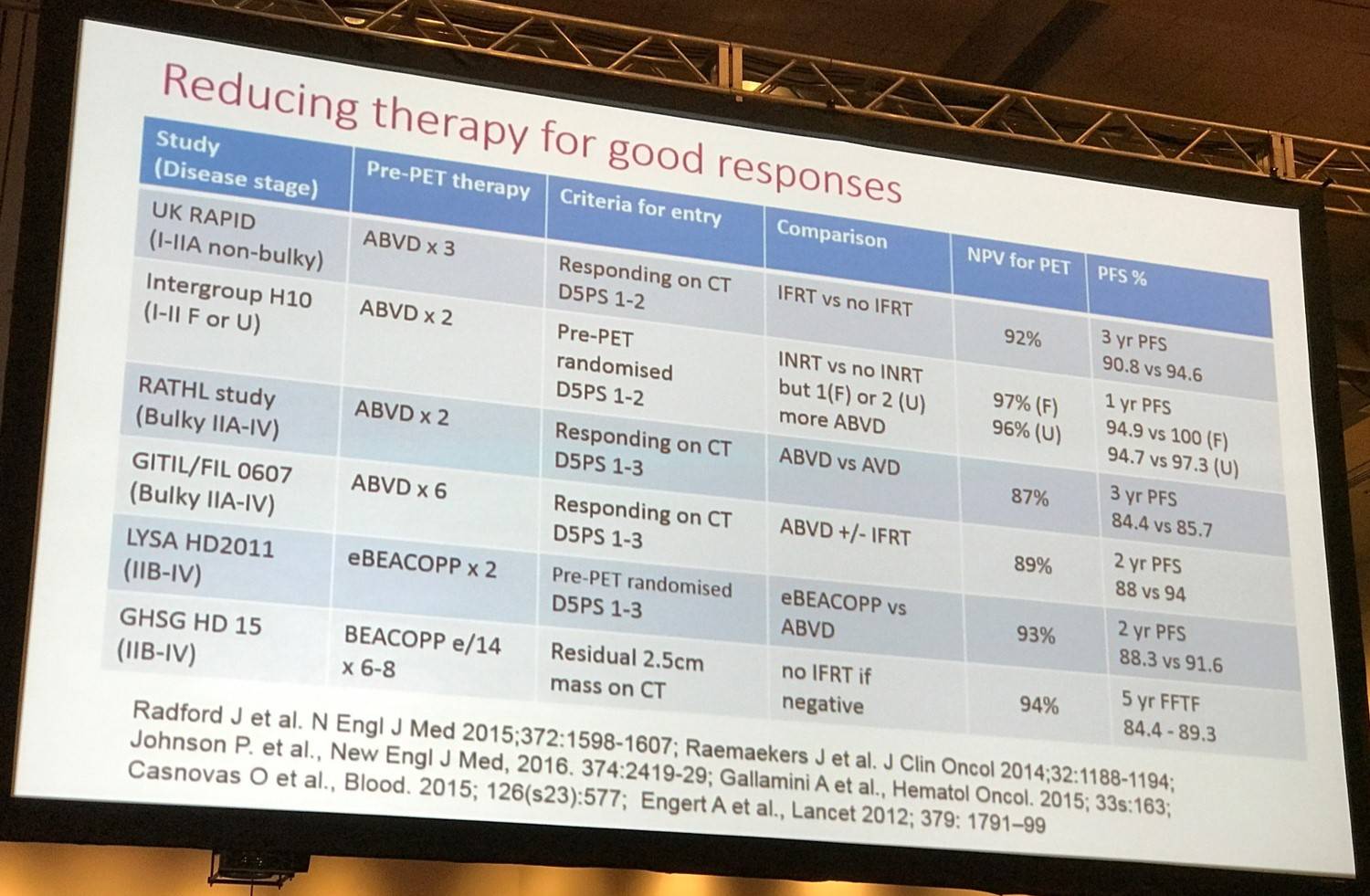
Increasing treatment after a positive PET scan in early stage disease
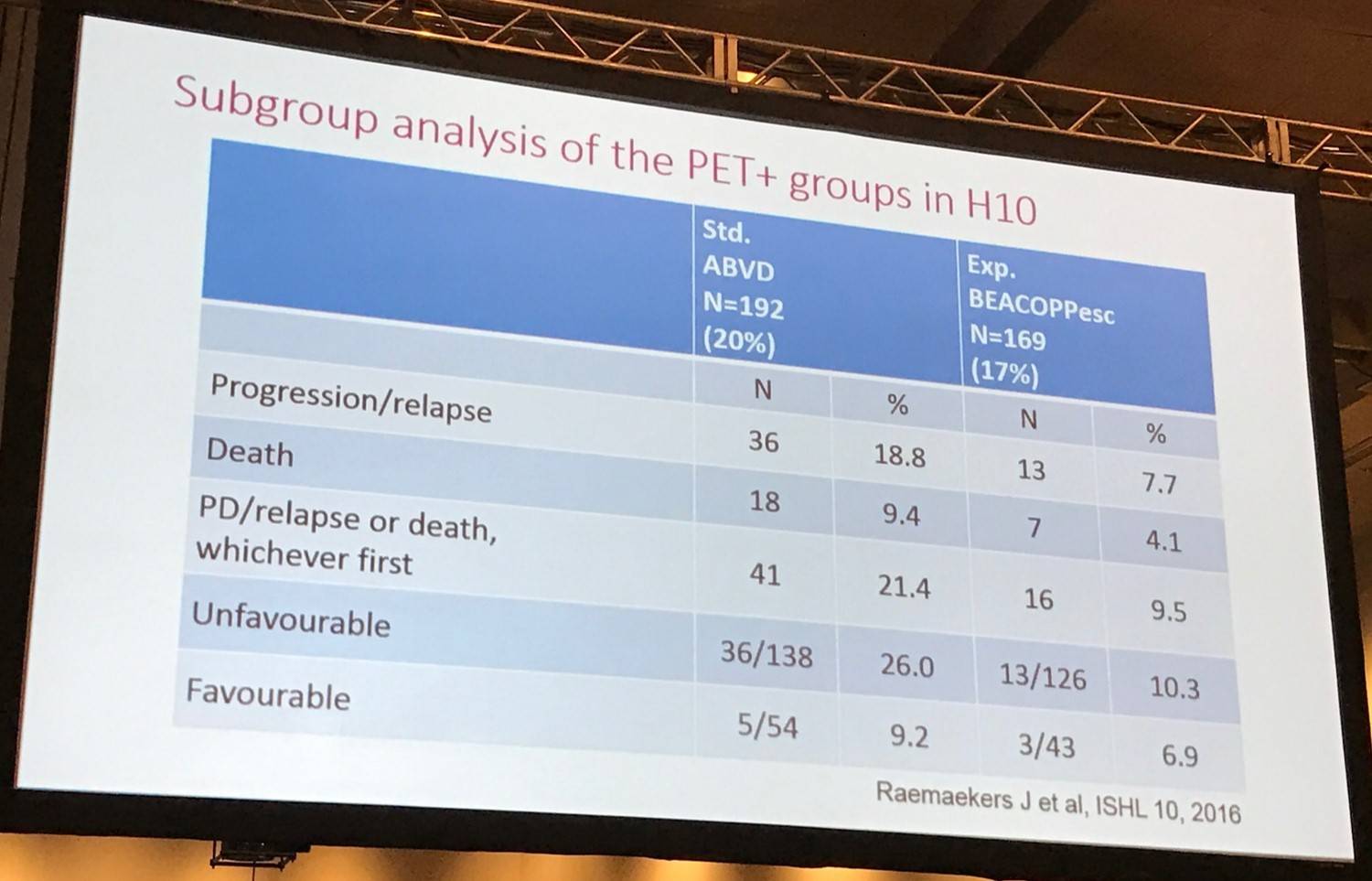
Dr Johnson then began to explore escalation of treatment after PET. Interim PET is effective at identifying patients who responded poorly to initial treatment. Escalating subsequent therapy is a potential way of improving their outcomes, supposing their disease is still sensitive to intensified chemotherapy or other treatment options such as radiotherapy. Many large prospective series have explore this theory but currently only one randomized trial has reported initial results in early stage disease: the EORTC H10 study.
- 19% of patients with positive PET scan
- Estimated 5-years PFS was increased from 77% with continued ABVD and INRT on the standard treatment arm to 91% with BEACOPPescalated and INRT in the PET-directed group (HR 0.42, P=.002)
- Trend towards improved OS
Increasing treatment after a positive PET scan in advanced stage disease
Many large-scale studies with very similar results have been reported which investigated treating PET-positive patients with BEACOPPescalated after 2 cycles of ABVD.

Comparing these studies to historical control studies in which the proportion of durable responses in PET-positive patients was only 15–30% indicates that escalating therapy is an effective way of improving outcomes for these patients. However, a PFS of 65–70% still leaves significant room for improvement.

Conclusion
Dr Johnson ended the talk by stating that response-adapted therapy using FDG-PET gives patients and their caregivers a chance to personalize their therapeutic approach and that he believes FDG-PET is currently the standard of care in HL.

Abstract
Treating Hodgkin lymphoma by using chemotherapy with or without radiotherapy is highly successful, with substantially fewer deaths from lymphoma than from other causes in recent studies of both early-stage and advanced-stage disease. Long-term toxicity is a major consideration in this context, and recent trials have used functional imaging with [18F]fluorodeoxyglucose (FDG) positron emission tomography early in the course of treatment (interim PET) to assess response and modulate subsequent therapy. In early-stage disease, this has allowed omission of consolidation radiotherapy after a good response with doxorubicin, bleomycin, vinblastine, and dacarbazine (ABVD) chemotherapy, and trials have shown that this can be one without detriment to overall survival, despite a small increase in rates of recurrence of ~5%. Conversely, escalation to more intensive chemotherapy with bleomycin, etoposide, doxorubicin, cyclophosphamide, vincristine, procarbazine, and prednisone (BEACOPP) for those with positive interim PET scans seems to be an effective strategy with improved disease control. In advanced-stage disease, several groups have elected to start treatment with ABVD and escalate to BEACOPP or myeloablative therapy for patients who remain PET positive after 2 cycles, which gives rates of disease control of ~65%. De-escalation by omission of bleomycin and consolidation radiotherapy after a negative interim PET scan seems safe with no increase in recurrence rate, but the performance of interim PET after ABVD is suboptimal, especially for those with very advanced disease at presentation; recurrence rates after a negative scan are ~15%. The negative predictive value of PET is higher after escalated BEACOPP chemotherapy, and the approach of initially treating with BEACOPP and de-escalating to ABVD for those with negative interim PET scans shows promising early results. Response-adapted therapy had yielded important results for patients with Hodgkin lymphoma and is becoming established as a standard approach.
- Johnson P.M. Response Adapted Frontline Therapy in Hodgkin Lymphoma: Are We There Yet? 2016 December 3; Educational Session Hodgkin Lymphoma: Treatment Today and the Challenge for Tomorrow. ASH 58th Annual Meeting and Exposition, San Diego, CA.

Understanding your specialty helps us to deliver the most relevant and engaging content.
Please spare a moment to share yours.
Please select or type your specialty
 Thank you
Thank youRelated articles
Newsletter
Subscribe to get the best content related to lymphoma & CLL delivered to your inbox








