All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a Healthcare Professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The Lymphoma Hub uses cookies on this website. They help us give you the best online experience. By continuing to use our website without changing your cookie settings, you agree to our use of cookies in accordance with our updated Cookie Policy
Introducing

Now you can personalise
your Lymphoma Hub experience!
Bookmark content to read later
Select your specific areas of interest
View content recommended for you
Find out moreThe Lymphoma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Lymphoma Hub cannot guarantee the accuracy of translated content. The Lymphoma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
ASH 2016 | Brentuximab vedotin plus nivolumab is well tolerated in patients with R/R HL: preliminary safety and response data of phase I E4412 trial
Bookmark this article
On 3–6 December 2016, the Annual Meeting & Exposition of the American Society of Hematology (ASH) took place in San Diego, CA.
On Monday 5th December, an oral abstract session was held between 4:30pm and 6:00pm in the “Hodgkin Lymphoma and T/NK Cell Lymphoma – Clinical Studies Program: Oral and Poster Abstracts” category. This session was moderated by Stephen Ansell, MD, PhD, of the Mayo Clinic and Anas Younes, MD, of the University of Texas M.D. Anderson Cancer Center.
Abstract #1106 was presented during this session, titled “A Phase I Study with an Expansion Cohort of the Combination of Ipilimumab and Nivolumab and Brentuximab Vedotin in Patients with Relapsed/Refractory Hodgkin Lymphoma: A Trial of the ECOG-ACRIN Cancer Research Group (E4412 Arms D and E)” by Catherine S. Diefenbach, MD, from NYU Perlmutter Cancer Center, NYU School of Medicine, New York, and colleagues.
At the data cut-off of 20th July 2016, 10 patients (1 ineligible) with confirmed R/R HL had been treated with brentuximab vedotin and nivolumab. The data reported in this presentation concerned the dose escalation population in Arm D (dose level 1: nivo 3mg/kg and BV 1.2mg/kg; n=3) and Arm E (dose level 2: BV 1.8mg/kg; n=7).
Safety
- Overall safety profiles show that brentuximab vedotin plus nivolumab was well tolerated
- In Arm E, one patient experienced a DLT: grade 3 pneumonia with grade 3 dyspnea and hypoxia, and grade 3 typhilitis (made full recovery, discontinued further therapy)
- No further DLTs noted, only 2 further grade 3 AEs were reported: rash and puritis
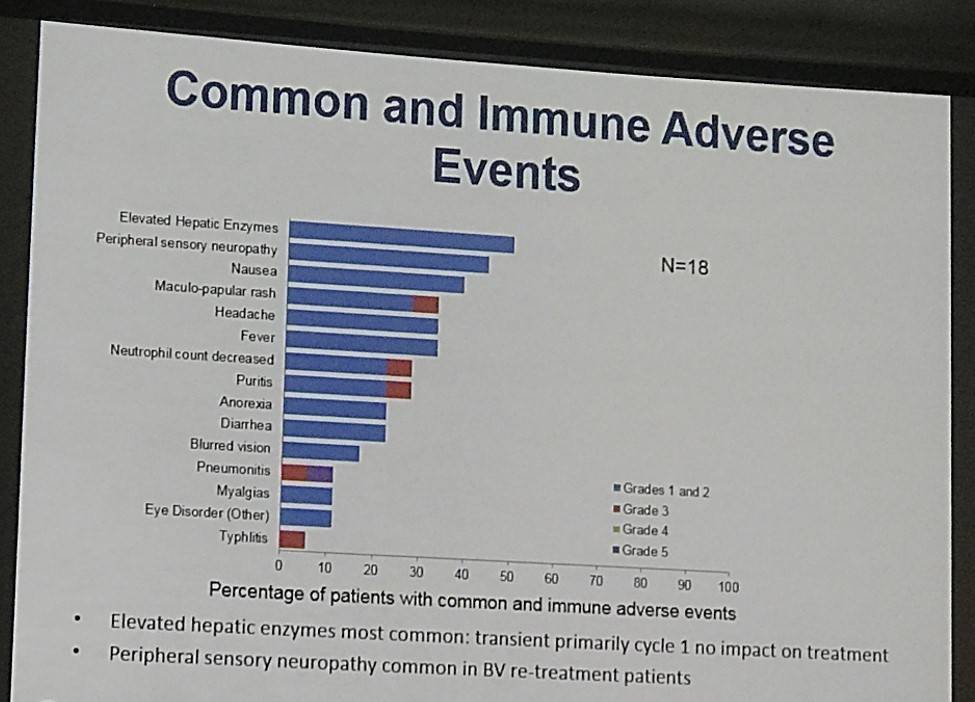
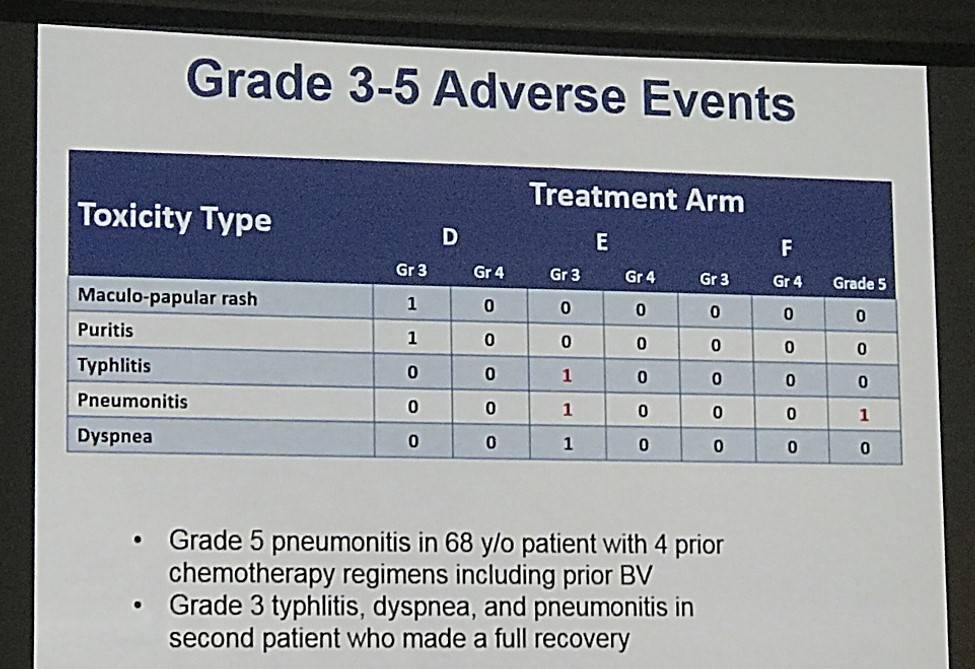
Response
- Eight of the 10 patients were evaluable
- ORR = 100%
- CR = 66% (8/12; 95% CI, 0.25–0.91)

- Two patients who had previously been treated with BV both achieved CRs
- PFS to date = 100%, median follow-up = 0.39 years
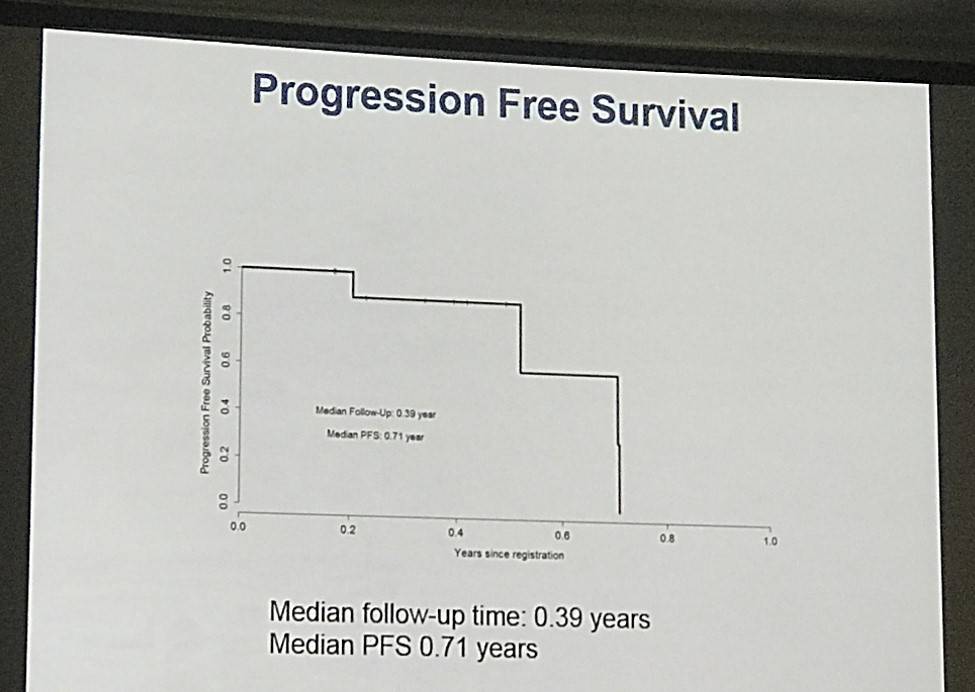
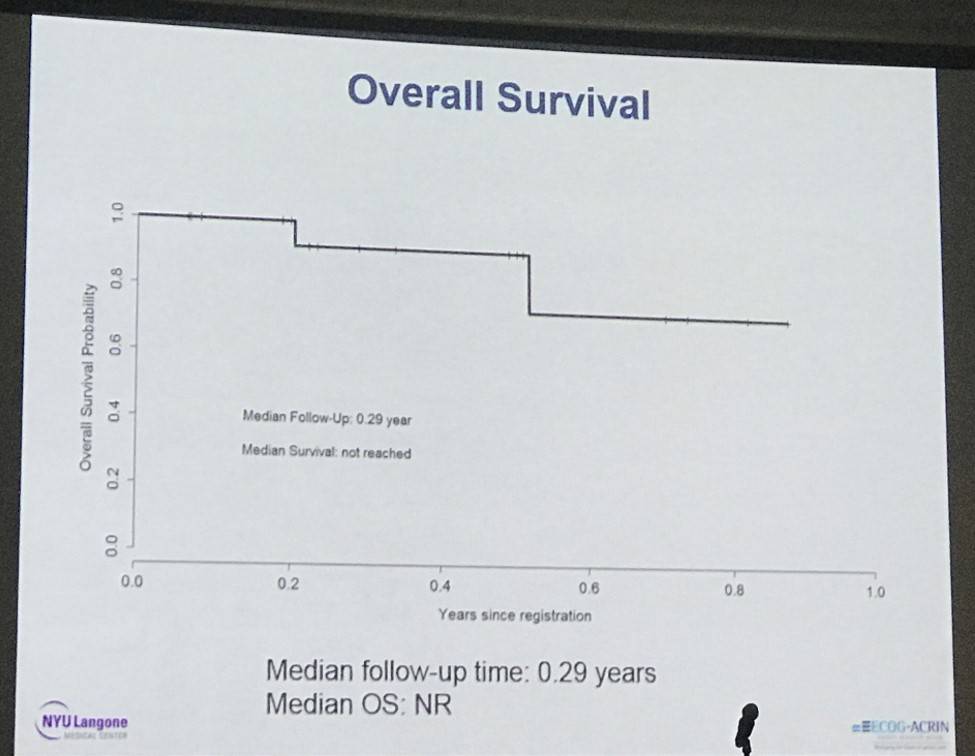
This is the first reported study of combining nivolumab with brentuximab vedotin. It was found this combination appears well tolerated. Further optimization of this combination strategy is planned; accrual is ongoing for patient cohorts receiving BV+nivo and BV+ipilimumab+nivo.

Abstract:
Background: Despite advances in chemotherapy, relapsed/refractory (R/R) Hodgkin lymphoma (HL) remains a significant clinical challenge and an unmet medical need. HL has a unique biology in which a small number of malignant Hodgkin Reed-Sternberg (HRS) cells propagate an immunosuppressive microenvironment. We hypothesized that using immune checkpoint inhibitor therapy to activate the immune cells in the tumor microenvironment, and concurrently targeting HRS cells with the CD30 antibody-drug conjugate brentuximab vedotin (BV), could overcome tumor cell resistance. E4412 is a Phase 1 ECOG-ACRIN sponsored study of the combination of BV and ipilimumab (IPI) and nivolumab (Nivo) in patients with R/R HL. Here we present the preliminary safety and response data on the patients treated with BV + Nivo (Arms D and E).
Methods: Patients with confirmed R/R HL were treated with Nivo 3mg/kg and BV 1.2mg/kg (Arm D: Dose Level 1) or 1.8mg/kg (Arm E: Dose Level 2) in dose escalation with a 3+3 design and an expansion cohort (Arm F) of 9 patients. BV and Nivo are given every 21 days for 16 cycles; Nivo may be continued for an additional year (total 2 years of Nivo therapy). Dose limiting toxicity (DLT) was defined for purposes of dose escalation within the first cycle of therapy. Patients are followed for toxicity up to 30 days beyond their last treatment.
Results: As of 7/20/2016 10 patients (1 ineligible) have been treated with BV + Nivo. We report the data on the dose escalation population: 3 patients: Arm D, 7 patients: Arm E. Data will be updated to include the full BV + Nivo dose escalation and dose expansion cohorts (Arms D, E & F) (N = 19) by the time of the Annual Meeting. Median age was 46; (range: 25-53). Six patients were male. Patients were heavily pretreated with a median of 3 prior therapies. Six patients had prior SCT (5 autologous, 1 allogeneic); 2 patients had prior treatment with BV.
Safety: Ten of 10 patients are evaluable for safety. Overall safety profiles show that the regimen of BV + Nivo was extremely well tolerated. One patient out of 7 in dose level 2 experienced a DLT (pneumonitis grade 3 with grade 3 dyspnea and hypoxia, and typhilits grade 3), this patient had received a prior SCT, and made a full recovery from his toxicities. Per protocol he discontinued further therapy. No other DLTs were noted for the remaining 9 patients. Common and relevant toxicities considered at least possibly related to drug during any cycle of treatment are shown in Table 1. There were no Grade 4 treatment related adverse events (AEs). The only additional grade 3 AEs noted were one each grade 3 rash and puritis. The most common grade 1-2 treatment related adverse events were: transaminitis (9), peripheral sensory neuropathy (6), and rash (3). Other AEs of interest included: diarrhea (3), blurry vision (2), and myalgias (2). One grade 1-2 infusion reaction was noted, this patient was able to receive subsequent therapy with pre-medication.
Response: Eight of 10 patients are evaluable for response. One patient is not yet evaluable. For the 8 evaluable patients the overall response rate (ORR) for the combination of BV + Nivo was 100%, with a CR rate of 62.5% (5/8), with 95% CI of 0.25-0.91. One patient was ineligible for laboratory studies that were out of range at screening, however this patient continues on therapy, and also had a response. One of 5 CRs occurred at in dose level 1 (BV 1.2 mg/kg). Both patients who had been treated with prior BV achieved CRs. The progression free survival (PFS) to date is 100% with a median follow-up of 0.3 years.
Conclusion: In this first reported study of the combination of the checkpoint inhibitor Nivo and the ADC BV therapy was well tolerated with one DLT, and primarily grade 1 and 2 toxicities. In a heavily pretreated patient population, 20% of whom had had prior BV and 60% of whom were s/p ASCT, the ORR of 100% and CR rate of 62.5% suggests a deepening of response compared to either therapy alone. Optimization of this combination strategy is planned with ongoing accrual to cohorts receiving BV + Nivo, and BV + Ipi + Nivo. Data will be updated to include the full BV + Nivo cohort (N = 19) by the time of the Annual Meeting.
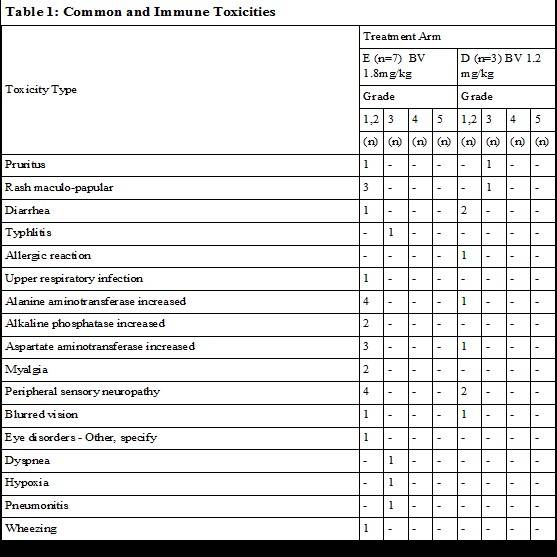
- Diefenbach C.S. et al. A Phase I Study with an Expansion Cohort of the Combination of Ipilimumab and Nivolumab and Brentuximab Vedotin in Patients with Relapsed/Refractory Hodgkin Lymphoma: A Trial of the ECOG-ACRIN Cancer Research Group (E4412 Arms D and E). Oral Abstract #1106: ASH 58th Annual Meeting and Exposition, San Diego, CA.

Understanding your specialty helps us to deliver the most relevant and engaging content.
Please spare a moment to share yours.
Please select or type your specialty
 Thank you
Thank youNewsletter
Subscribe to get the best content related to lymphoma & CLL delivered to your inbox








