All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a Healthcare Professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The Lymphoma Hub uses cookies on this website. They help us give you the best online experience. By continuing to use our website without changing your cookie settings, you agree to our use of cookies in accordance with our updated Cookie Policy
Introducing

Now you can personalise
your Lymphoma Hub experience!
Bookmark content to read later
Select your specific areas of interest
View content recommended for you
Find out moreThe Lymphoma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Lymphoma Hub cannot guarantee the accuracy of translated content. The Lymphoma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
ASH 2016 | Ibrutinib-Lenalidomide-Rituximab in Patients with Relapsed/Refractory Mantle Cell Lymphoma: First Results from the Nordic Lymphoma Group MCL6 (PHILEMON) Phase II Trial
Bookmark this article
The 58th Annual Meeting & Exposition of the American Society of Hematology’s (ASH) took place in San Diego, CA, and on December 3rd, Mats Jerkeman, from Skåne University Hospital, Lund, Sweden, presented results from a multi-center, open-label, phase-II trial which examined the safety and efficacy of a triple combination therapy of ibrutinib-lenalidomide-rituximab in R/R MCL patients. The inclusion criteria were MCL patients R/R after rituximab containing treatment, with measurable disease and WHO PS 0–3.
Highlights:
- Induction Treatment:
- Max 12, 28 day cycles of: 15mg lenalidomide daily (days 1-21), 560 mg ibrutinib daily (days 1-28), 375 mg/m2 rituximab day 1, cycle 1, then 1400mg S.C. or 375mg/m2V. (days 8, 15, 22 in cycle 1, then just day one in cycles 3, 5, 7, 9, 11)
- Maintenance Treatment:
- Patients in CR, PR, SD = 560mg ibrutinib daily, 1400mg S.C. or 375mg/m2V. on day one
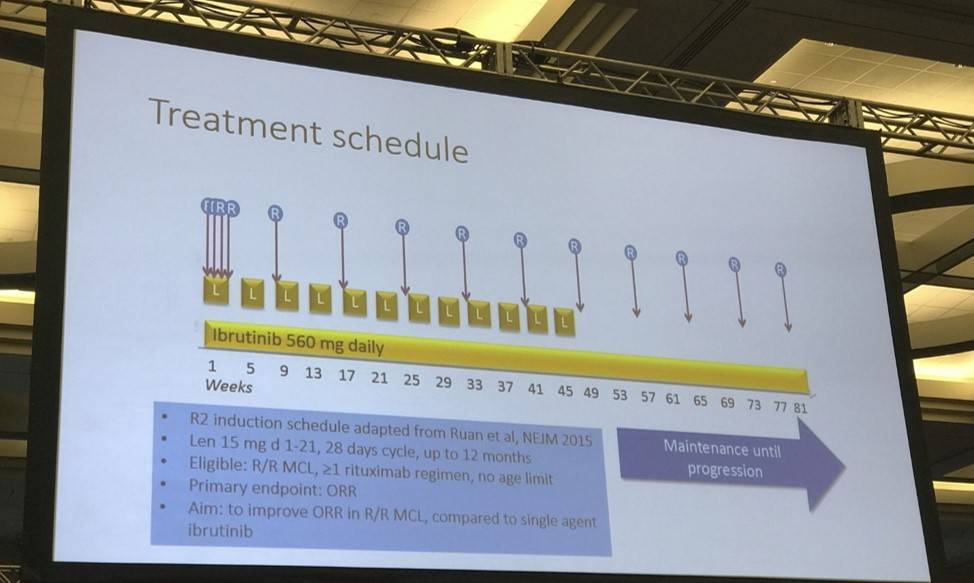
- Results:
- 50 pts over 10 centers in 4 countries
- 34% of pts discontinued treatment (17 total, 3 PD, 4 AE)
- Low grade hematological AEs, apart from neutropenia grade 3–4, in 10% of patients
- One patient died due to septic shock, potentially treatment related
- 29 evaluable pts at median follow-up of 7 months
- Median Progression Free Survival (PFS) at median follow-up of 8 months has not been reached
- ORR = 88%, CR 64%, PR 24%
- 13 MRD evaluable patients: 7/12 blood molecular remission, 7/13 bone marrow remission
- 8-month PFS in TP53 mutant vs no TP53 mutant ; 71% vs 72% (P = 0.43)
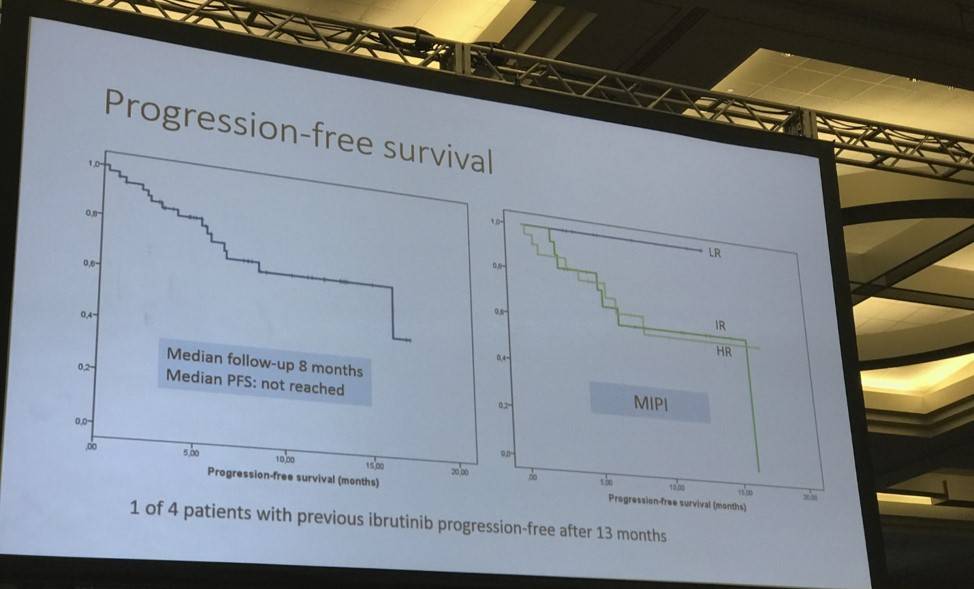
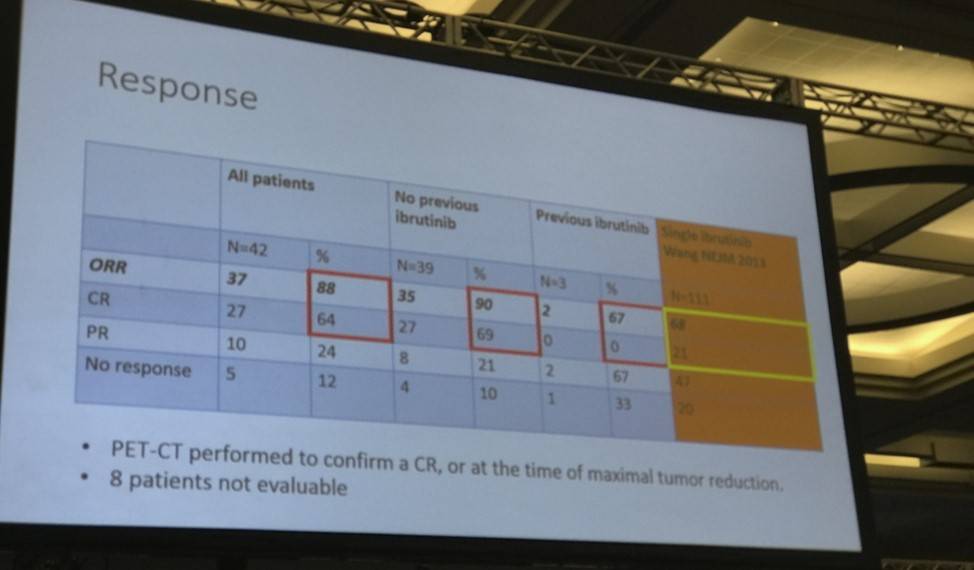
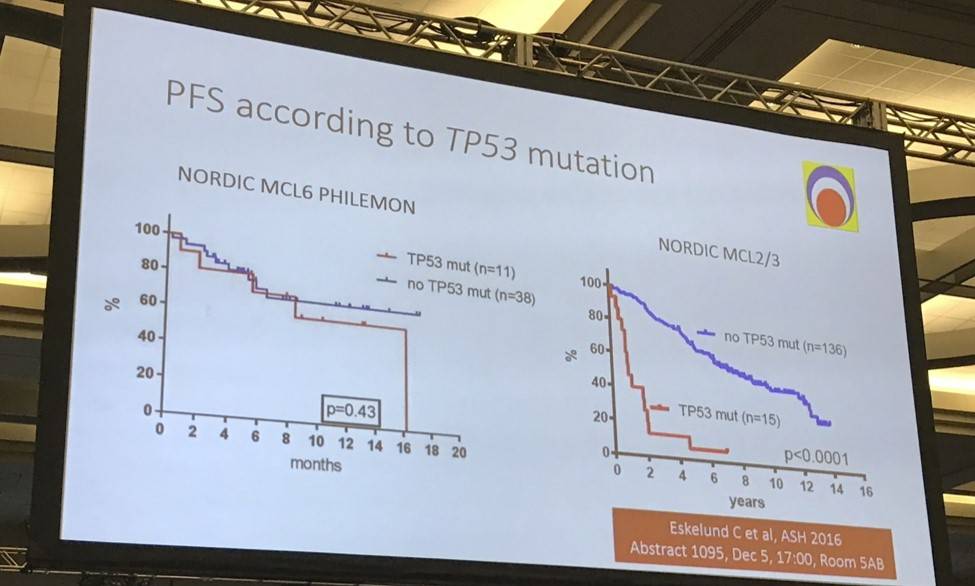
Mats Jerkeman ended his talk by concluding that this ibrutinib-lenalidomide-rituximab combination therapy, in R/R MCL patients was well-tolerated, active and associated with molecular remission via both blood and bone marrow assessment.

Abstract:
Introduction: In spite of improvements in treatment of mantle cell lymphoma (MCL) over the last 10-15 years, this is still considered an incurable lymphoma entity, and the majority of patients eventually relapse. Ibrutinib is a very active agent in MCL, with an overall response rate (ORR) of 68% as a single agent in the relapse situation. In vitro, ibrutinib has been shown to partially antagonize the activity of rituximab, by suppression of NK cell activity and subsequent ADCC. Lenalidomide, on the other hand, improves rituximab-induced ADCC. In this multi-centre open-label phase II trial, we evaluated safety and efficacy of this triplet combination in patients with relapsed or refractory MCL. A phase I trial with this combination has been performed in 22 patients with untreated follicular lymphoma (Alliance 051103). In this trial, rash was the most common adverse event (AE), occuring in 73% of pts, with grade 3 rash in 32%.
Methods: Eligibility criteria were: patients with MCL, relapsing after or refractory to at least one rituximab-containing chemotherapy regimen, WHO PS 0-3, and measurable disease. The primary endpoint was maximal overall response rate (ORR) measured with CT and PET/CT. Minimal residual disease (MRD) monitoring by PCR was performed during follow-up, according to EuroMRD criteria. Ion Torrent sequencing was performed on frozen tumor cells from bone marrow at time of relapse, including the following genes: ATM, CCND1, TP53, KMT2D, NOTCH1, NOTCH2, WHSC1 and BIRC3.
Treatment schedule: Induction phase: Up to twelve 28-day cycles with: Lenalidomide 15 mg p o daily, days 1-21, Ibrutinib 560 mg p o days 1-28, Rituximab 375 mg/m2 i v day 1 in cycle 1, then 1400 mg s c (or 375 mg/m2i v) days 8, 15 and 22 in cycle 1, then day 1 in cycles 3, 5, 7, 9 and 11. Maintenance phase: For patients in CR, PR or SD, not in need of other treatment. Given until progression, cycle duration 56 days. Ibrutinib: 560 mg p o days 1-56, 2. Rituximab 1400 mg s c (or 375 mg/m2i v) day 1 of each cycle.
Results: Accrual of 50 pts was completed in 12 months, June 2016, at 10 centres in Sweden, Norway, Denmark and Finland. The median age was 69.5 years, with a median MIPI score of 6.2. Patients had received a median of two previous regimens, four had progressed after single agent ibrutinib, and three had received prior allo-SCT. In total, 17/50 pts have discontinued treatment (n=9 due to PD, n=4 due to AE, n=2 withdrew consent, n=1 proceeded to alloSCT and n=1 due to other cause). Treatment emergent-AEs of any grade in ≥20% of patients were rash (24%) and fatigue (20%). Five pts (10%) experienced rash grade 3, mainly during cycle 1. Hematological toxicity was generally of low grade, apart from grade 3-4 neutropenia in 5 patients. One patient died due to possible treatment-related toxicity (septic shock). One event of laboratory tumor lysis syndrome was reported, and two events of atrial fibrillation, without reduction or discontinuation of ibrutinib. With a median follow up time of 7 months, 29 patients were evaluable for efficacy as of July 14, 2016. The ORR to date is 83% with 12 patients achieving CR (41%) and 12 PR (41%). Median duration of response and PFS has not been reached. One of three evaluable patients with progression on single agent ibrutinib responded with a PR, with ongoing response at 9 months. Of the 13 patients evaluable for MRD at 6 months, 7/12 patients have achieved molecular remission in blood and 7/13 in bone marrow.
Conclusions: So far, the combination of ibrutinib, lenalidomide and rituximab has been shown to be an active and well tolerated regimen in patients with R/R MCL, associated with molecular remission. Cutaneous toxicity was manageable, in contrast to what has been reported with a similar combination in untreated patients with follicular lymphoma. Up-dated results will be presented at the annual meeting, including data on mutational profile as biomarker for efficacy. This trial was registered at http://clinicaltrials.gov as NCT02460276.
- Jerkeman M. et al. Ibrutinib-Lenalidomide-Rituximab in Patients with Relapsed/Refractory Mantle Cell Lymphoma: First Results from the Nordic Lymphoma Group MCL6 (PHILEMON) Phase II Trial. 2016 December 3; Oral Abstract #148: ASH 58th Annual Meeting and Exposition, San Diego, CA.

Understanding your specialty helps us to deliver the most relevant and engaging content.
Please spare a moment to share yours.
Please select or type your specialty
 Thank you
Thank youRelated articles
Newsletter
Subscribe to get the best content related to lymphoma & CLL delivered to your inbox








