All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a Healthcare Professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The Lymphoma Hub uses cookies on this website. They help us give you the best online experience. By continuing to use our website without changing your cookie settings, you agree to our use of cookies in accordance with our updated Cookie Policy
Introducing

Now you can personalise
your Lymphoma Hub experience!
Bookmark content to read later
Select your specific areas of interest
View content recommended for you
Find out moreThe Lymphoma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Lymphoma Hub cannot guarantee the accuracy of translated content. The Lymphoma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
ASH 2016 | Pembrolizumab for Treatment of Relapsed/Refractory Mycosis Fungoides and Sezary Syndrome: Clinical Efficacy in a Citn Multicenter Phase 2 Study
Bookmark this article
The 58th Annual Meeting & Exposition of the American Society of Hematology’s (ASH) took place in San Diego, CA, and on December 3rd, Michael Khodadoust, MD, PhD, from Stanford University School of Medicine, CA, USA, presented the results of a Phase-II, single-arm, multi-center study into the clinical role of the immune checkpoint inhibitor pembrolizumab in the treatment of R/R mycosis fungoides and Sézary syndrome (MF/SS). The population in this study was 24 patients who had received at least one prior therapy.
Highlights:
- 96% pts had stage IIB or higher, 63% stage IVA SS
- Treatment: 2mg/kg every 3 weeks, for up to 2 years
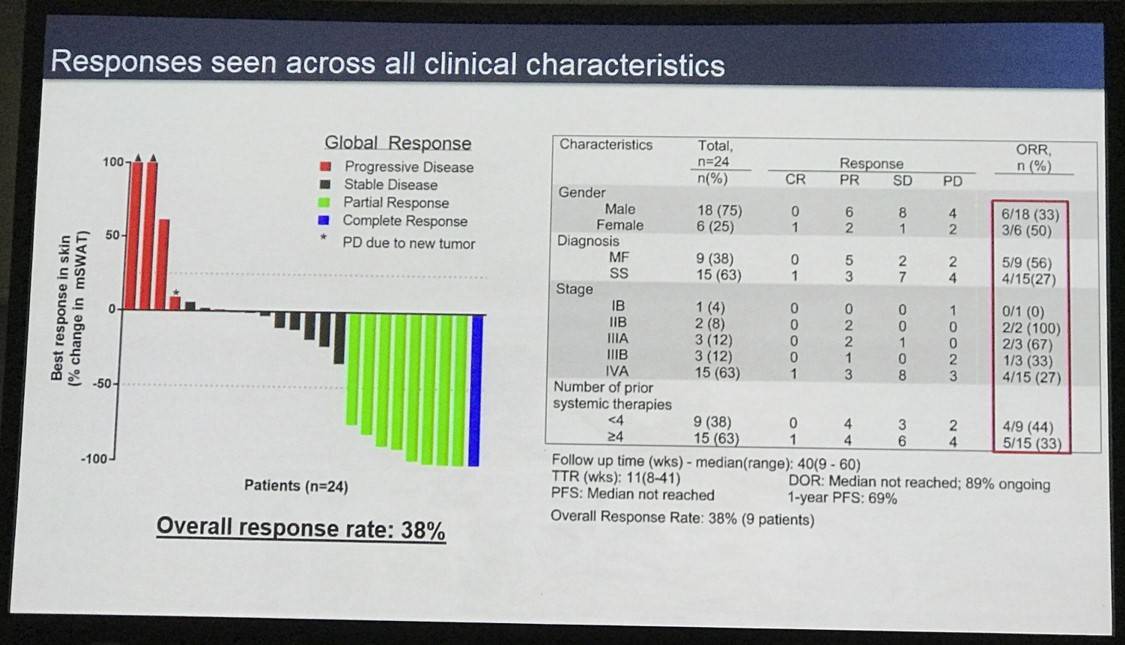
- ORR = 38% (1 CR, 8 PR, 9 SD)
- 1 year PFS = 69%
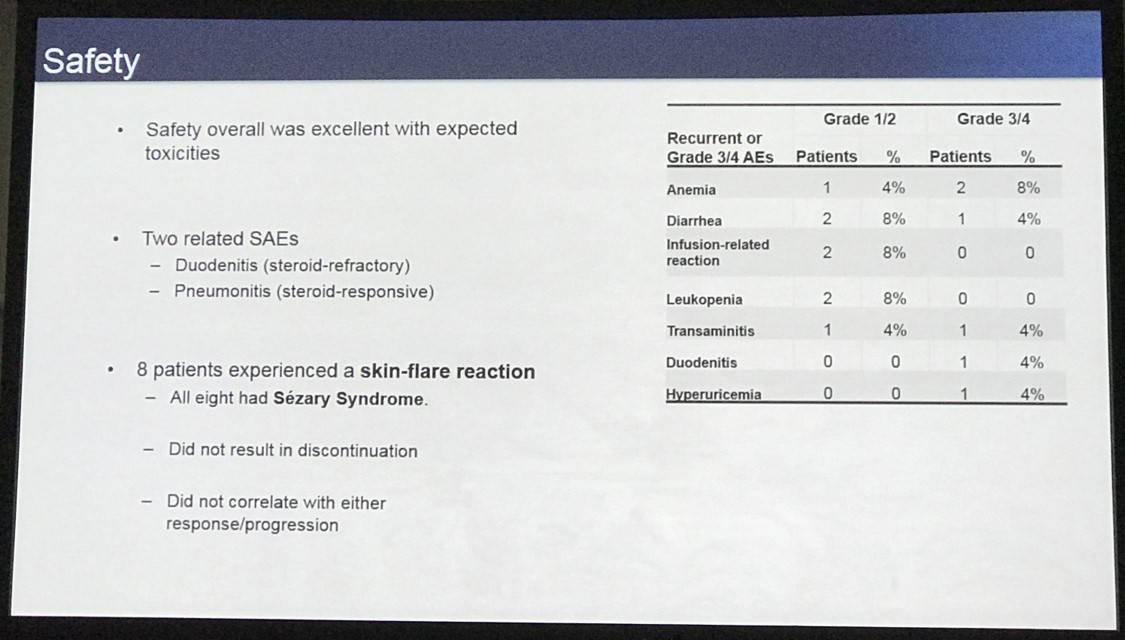
- Toxicities:
- As previously reported, plus immune-mediated skin flare (6pts/40% of SS patients)
- Two SAEs which were immune related
Michael Khodadoust concluded that pembrolizumab was active and well tolerated in patients with previously treated MF/SS. A further phase-II trial of pembrolizumab + interferon-gamma is being worked on.

Abstract:
Background: PD-1 and PD-L1/PD-L2 are expressed by malignant T-cells in mycosis fungoides (MF) and Sézary syndrome (SS). PD-1 is additionally expressed by tumor-infiltrating cytotoxic T-cells and PD-L1 is expressed by macrophages and other stromal components of the tumor microenvironment in these diseases. Moreover, reports of 9p24.1/PD-L2 translocation and CTLA4-CD28 fusion events in MF/SS support a genomic basis for immune evasion. Here, we explore the clinical activity of pembrolizumab, an immune checkpoint inhibitor of the PD-1/PD-L1 axis, in MF/SS.
Methods: Patients (pts) with MF/SS stages IB-IV treated with at least 1 prior systemic therapy were enrolled in this phase 2, single-arm study coordinated by the Cancer Immunotherapy Trials Network (CITN). A Simon two-stage design was applied where stage 2 is initiated if 1 of 9 pts had an objective response. An additional 15 pts were planned in stage 2. Pembrolizumab was administered at 2 mg/kg every 3 weeks and treatment was allowed up to 2 years. The primary endpoint was overall response rate (ORR) as determined by the consensus global response criteria. Secondary endpoints were safety/tolerability, time to response (TTR), duration of response (DOR) and progression-free survival (PFS). Correlative biomarker studies included immunohistochemistry (IHC) staining for PD-L1, PD-L2, and multiple immune subsets as well as serum analysis of 62 cytokines and chemokines. Phenotypic and functional profiling of malignant and non-malignant immune cells will be performed by flow cytometry and mass cytometry (CyTOF).
Results: The study completed enrollment and all 24 patients received at least one dose of pembrolizumab. Median age was 67 (range 44-85); 18 were male. Patients were advanced stage with 23 patients (96%) stage IIB or higher, including 15 patients (63%) with stage IVA SS. Most pts were heavily treated with a median of 4 prior systemic therapies (range 1-11). The median follow-up time was 40 weeks (range 9-60 weeks). The objective response rate (ORR) was 38% with 1 complete response (CR) and 8 partial responses (PR). Of the responding pts, 6 pts had 90% or greater improvement in skin disease as measured by mSWAT. An additional 9 pts (38%) had stable disease (SD). The median TTR was 11 weeks (range 8-41 weeks). Responses were durable with 8 of 9 (89%) responses currently ongoing at a median of 32 weeks of duration (4-46). The median PFS has not yet been reached, and the one-year PFS was 69%. There was no significant association between response and clinical characteristics including stage, disease type (MF vs. SS), and number of prior therapies, nor with skin tissue expression of PD-1, PD-L1, PD-L2, or infiltrating CD8+ T-cells as determined by IHC. Planned additional correlatives including CyTOF profiling, gene expression profiling, T cell receptor high throughput sequencing, multiplexed ion beam imaging (MIBI), and whole exome sequencing will explore potential predictive biomarkers of response.
Adverse events (AE) were consistent with those seen in prior studies of pembrolizumab with the exception of an immune-mediated skin flare reaction seen in 6 pts (2 grade 2 and 4 grade 3). Skin flares occurred exclusively in patients with SS (6/15; 40%) and were associated with lower serum levels of the cytokines IL-7 and SCF prior to pembrolizumab treatment (p=0.01 and p=0.02 respectively, n.s. by Bonferroni correction). Pts with the skin flare reaction experienced increases in serum IFN-gamma, IL-12p40, IL-15, LIF, G-CSF, and CCL4 following treatment. There were two treatment related serious adverse events (SAE), both immune related. One pt experienced grade 2 pneumonitis which resolved with systemic corticosteroids. Another patient experienced grade 3 diarrhea secondary to steroid-refractory duodenitis.
Conclusions: Pembrolizumab has significant clinical activity in pts with previously treated MF/SS. Responses were durable and were not associated with any identifiable clinical or pathologic characteristics. Treatment was well tolerated with a toxicity profile consistent with prior pembrolizumab studies, though 40% of pts with SS developed a notable skin flare reaction. These findings support further study of PD-1 blockade in the treatment of MF and SS. A phase 2 trial of pembrolizumab in combination with interferon-gamma is being developed based on these results.
- Khodadoust M. et al. Pembrolizumab for Treatment of Relapsed/Refractory Mycosis Fungoides and Sezary Syndrome: Clinical Efficacy in a Citn Multicenter Phase 2 Study. 2016 December 3; Oral Abstract #181: ASH 58th Annual Meeting and Exposition, San Diego, CA.

Understanding your specialty helps us to deliver the most relevant and engaging content.
Please spare a moment to share yours.
Please select or type your specialty
 Thank you
Thank youRelated articles
Newsletter
Subscribe to get the best content related to lymphoma & CLL delivered to your inbox








