All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a Healthcare Professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The Lymphoma Hub uses cookies on this website. They help us give you the best online experience. By continuing to use our website without changing your cookie settings, you agree to our use of cookies in accordance with our updated Cookie Policy
Introducing

Now you can personalise
your Lymphoma Hub experience!
Bookmark content to read later
Select your specific areas of interest
View content recommended for you
Find out moreThe Lymphoma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Lymphoma Hub cannot guarantee the accuracy of translated content. The Lymphoma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
ASH 2016 | Lenalidomide is an efficient option for maintenance treatment: Interim analysis of the CLLM1 study
Bookmark this article
The American Society of Hematology’s (ASH) the 58th Annual Meeting & Exposition took place on 3–6 December 2016 in San Diego, CA. On Saturday 3rd December, an oral abstract session was held between 4:00pm and 5:30pm in the “CLL: Therapy, Excluding Transplantation Program” category. This session was moderated by Tait D. Shanafelt, MD, of the Mayo Clinic, and John F. Seymour, MB, BS, PhD, FRACP, of the Peter MacCallum Cancer Centre and Royal Melbourne Hospital.
Abstract #229 was presented during this session, titled “Lenalidomide Maintenance after Front Line Therapy Substantially Prolongs Progression Free Survival in High Risk CLL: Interim Results of a Phase 3 Study (CLL M1 Study of the German CLL Study Group)” by Anna Maria Fink, MD, from the Department I of Internal Medicine and Center of Integrated Oncology Cologne-Bonn, German CLL Study Group, University Hospital of Cologne, Cologne, Germany, and colleagues.
The primary endpoint of the CLLM1 study (NCT01556776), aiming to investigate lenalidomide as maintenance therapy versus placebo in a double-blind fashion, was Progression Free Survival (PFS) by independent review. This interim analysis was performed after 20% (24 events) of the calculated PFS events and 89/200 planned patients were randomized. Of the 89 randomized patients, 60 were administered lenalidomide and 29 received placebo. Median age of the randomized patients was 64 (32–80) and 85.2% were male. At randomization, 37% patients had a high and 63% of patients had intermediate Minimal Residual Disease MRD) level, respectively.
Highlights:
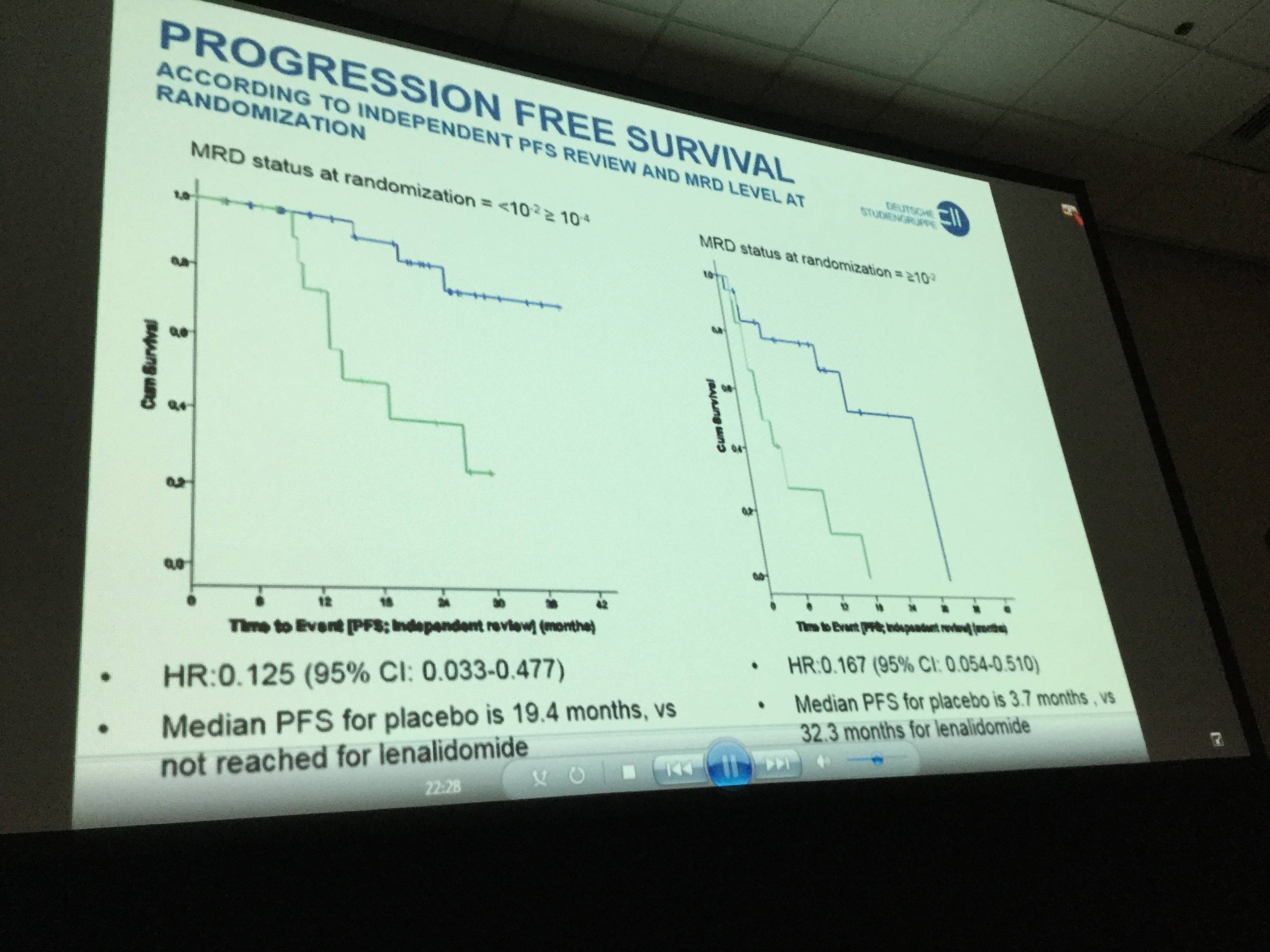
- MRD status at randomization <10-2 ≥ 10-4:
- PFS Hazard Ratio (HR) = 0.125 (95% CI:0.033-0.477)
- Median PFS placebo = 19.4 months, not reached for lenalidomide
- MRD status at randomization ≥ 10-2:
- PFS HR = 0.167 (95% CI: 0.054-0.510)
- Median PFS placebo = 3.7 months vs. 32.3 months lenalidomide
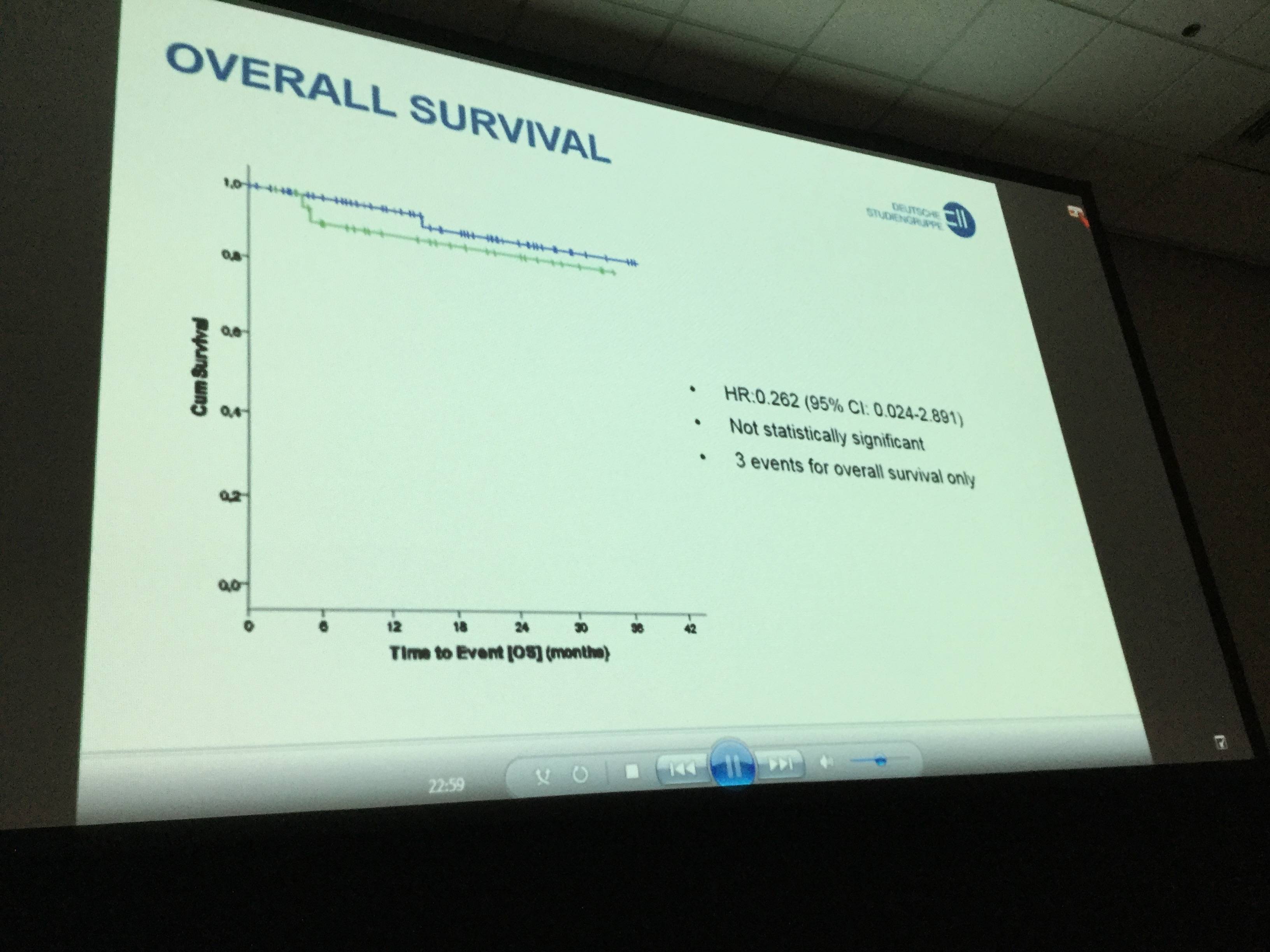
- OS differences between placebo and lenalidomide were not significantly different (HR:0.262 [95% CI:0.024-2.891])
- Conversion from MRD positivity to negativity has been reported in the lenalidomide treatment group
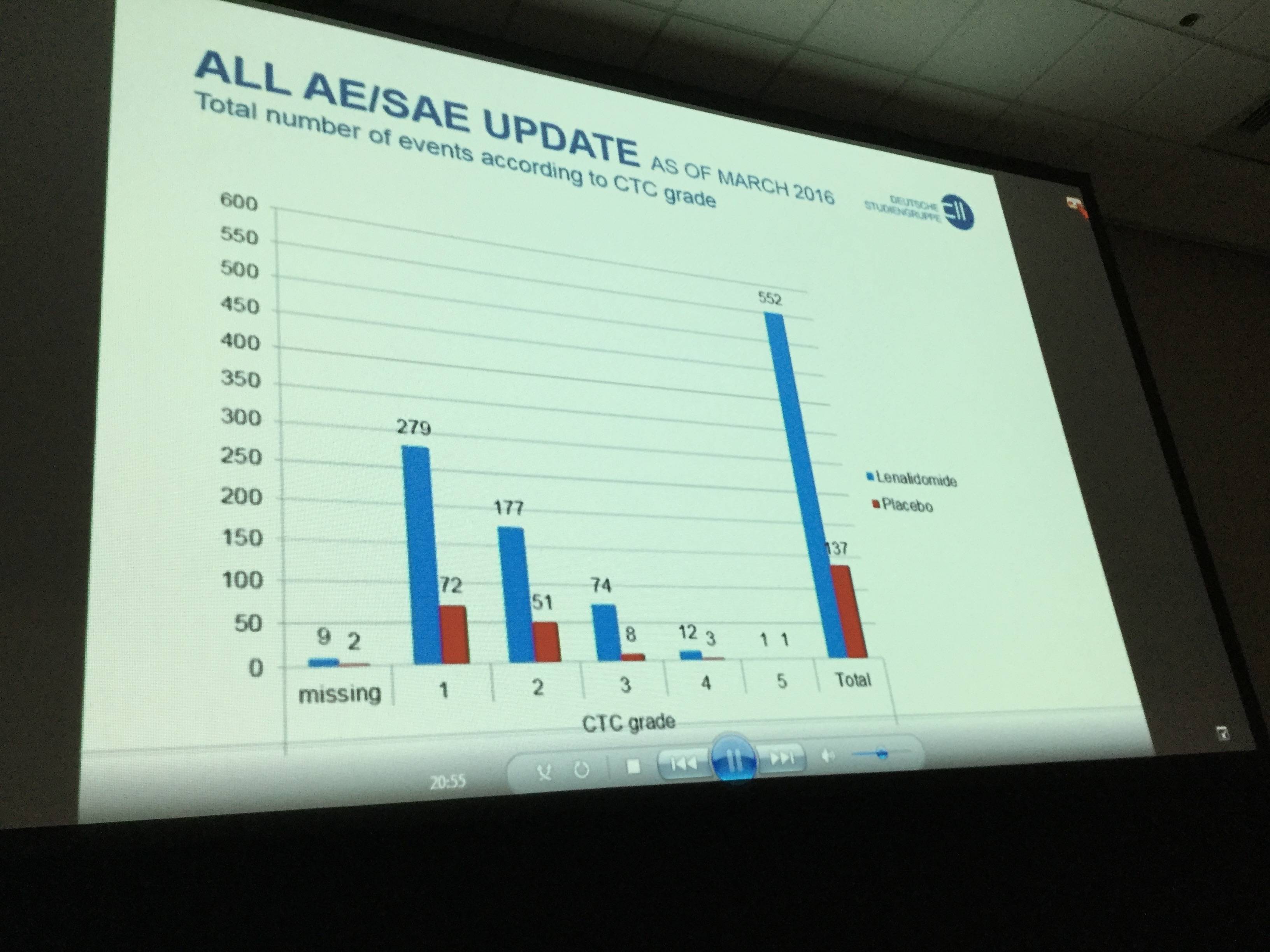
- Discontinuation of treatment occurred in 24 and 21 patients in the lenalidomide and placebo groups, respectively, due to AEs (18 vs 6), disease progressions (4 vs 13), withdrawal of consent (4 vs 2) and other reasons (2 vs 2)
- Neutropenia (30.4% vs 4%) and gastrointestinal disorders (55.4% vs 27.6%) were more common in the lenalidomide treatment arm than in the placebo arm
- Vascular disorders (14.3% vs 2%) and infections (50% vs 62.1%) did not increase in the lenalidomide patient group compared to placebo
- So far, 3 deaths have been reported: 1 in the lenalidomide arm (Acute Lymphoblastic Leukemia) and 2 in the placebo group (1 Progressive Multifocal Leukoencephalopathy and 1 Richter’s Syndrome)

This presentation concluded by stating that lenalidomide is an efficient option for maintenance treatment in patients with high-risk CLL after chemoimmunotherapy. Lenalidomide significantly prolonged PFS, resulting in a relative risk reduction for progression of >80%, although this has not translated into reported overall survival yet. Furthermore, this data confirm the prognostic values of MRD risk stratification in CLL.
Abstract
Introduction: The combined use of genetic markers and minimal residual disease (MRD) assessment identifies CLL patients (pts) with a poor outcome after front line chemoimmunotherapy. The CLLM1 study was initiated to evaluate lenalidomide as a maintenance treatment versus (vs) placebo in a double-blind fashion. The primary endpoint was progression free survival (PFS) by an independent review.
Methods: Pts who achieved at least a partial response after at least 4 cycles of front line chemoimmunotherapy were defined as high risk for progression if they had MRD levels of ≥10-2 or MRD levels of ≥10-4 to <10-2combined with either an unmutated IGHV gene status, del(17p) or TP53 mutation at baseline. After 2:1 randomization, treatment with lenalidomide or placebo started with 5 mg daily in the first cycle, and was subsequently escalated to the target dose of 15 mg in the 7th cycle. Further possible escalation was guided by six-monthly MRD assessments. If well tolerated, the study drug was administered until disease progression.According to their risk for thromboembolic events pts received either low dose aspirin daily, or appropriate anti-coagulation prophylactic therapies. Pts were followed for progression monthly. This formal interim analysis was conducted after 20% (24 events) of the calculated PFS events and 89 pts of the planned 200 pts were randomized. PFS was evaluated at the significance level determined using the Hwang-Shih-DeCani spending function (including stopping boundaries for both futility and efficacy). The trial was registered with clinicaltrials.gov (NCT01556776).
Results: A total of 468 pts from 62 sites in five countries (Austria, Germany, Italy, Netherlands and Spain) were screened for the study. 379 were not eligible, 347 because of MRD-negativity/low risk after frontline treatment and 32 because of progression between end of frontline and screening (11), withdrawal of consent (15) and other reasons (6). Out of 89 randomized pts, 60 received lenalidomide and 29 placebo. Randomized pts had a median age of 64 (range, 32-80), a median CIRS Score of 2 (range, 0-6), and 85.2% were male. 11.4% /20.5% of the pts had a 17p deletion/TP53 mutation, and 90.2% of the pts had an unmutated IGHV gene status at baseline. At randomization, 37% of pts had a high and 63% an intermediate MRD level, respectively. At data cut off, pts had received a median of 10 (range 0-42) treatment cycles [lenalidomide, 11 (0-42); placebo, 9 (1-35)]. Treatment was discontinued in 27 pts on lenalidomide and in 21 pts on the placebo arm due to adverse events (17 vs 6 pts), disease progression (4 vs 13 pts), withdrawal of consent (4 vs 2 pts) and other reasons (2 vs 2 pts). Treatment with lenalidomide was more frequently associated with neutropenia (30.4% vs 3.4%), gastrointestinal disorders (55.4% vs 27.6%), nervous system disorders (30.4% vs 13.8%), respiratory disorders (35.7% vs 13.8%) and skin disorders (60.7% vs 27.6%). Infections (50% vs 62.1%) and vascular disorders (14.3% vs 17.2%) were not increased in the lenalidomide compared to the placebo arm. Three deaths have been observed so far, one in the lenalidomide arm (acute lymphoblastic leukemia) and two in the placebo arm (1 progressive multifocal leukoencephalopathy; 1 Richter’s syndrome). After a median observation time of 17.7 months, the hazard ratio for PFS was 0.198 with 95% confidence interval (CI) 0.083 to 0.475 (stratified by MRD level at randomization). The median PFS in the placebo arm was 14.6 months, and was not reached in the lenalidomide arm (see figure 1). The one-sided p-value from the stratified log-rank test was 0.000059/2, thus smaller than the determined efficacy boundary 0.0006 derived from the Hwang-Shih-DeCani spending function based on 24 PFS events. Conversion to MRD negativity has been observed only in the lenalidomide arm, data will be presented at the meeting.
Conclusion: Lenalidomide is a feasible and efficacious maintenance option for high risk CLL after chemoimmunotherapy and substantially prolonged PFS in high risk CLL patients resulting in a relative risk reduction for progression of more than 80%. An independent data monitoring committee assessed the results as being robust and reliable and recommended unblinding of the study as well as continuing treatment with lenalidomide. The PFS observed in the placebo arm independently confirms the prognostic significance of the MRD based risk assessment model, which might be used in future trials.
- Fink A.M. et al. Lenalidomide Maintenance after Front Line Therapy Substantially Prolongs Progression Free Survival in High Risk CLL: Interim Results of a Phase 3 Study (CLL M1 Study of the German CLL Study Group). Oral Abstract #229: ASH 58th Annual Meeting and Exposition, San Diego, CA.

Understanding your specialty helps us to deliver the most relevant and engaging content.
Please spare a moment to share yours.
Please select or type your specialty
 Thank you
Thank youRelated articles
Newsletter
Subscribe to get the best content related to lymphoma & CLL delivered to your inbox








