All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a Healthcare Professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The Lymphoma Hub uses cookies on this website. They help us give you the best online experience. By continuing to use our website without changing your cookie settings, you agree to our use of cookies in accordance with our updated Cookie Policy
Introducing

Now you can personalise
your Lymphoma Hub experience!
Bookmark content to read later
Select your specific areas of interest
View content recommended for you
Find out moreThe Lymphoma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Lymphoma Hub cannot guarantee the accuracy of translated content. The Lymphoma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
ASH 2016 | Lenalidomide maintenance in R-CHOP treated elderly patients with DLBCL
Bookmark this article
The 58th Annual Meeting & Exposition of the American Society of Hematology’s (ASH) took place in San Diego, CA, on December 3–6, 2016. On Sunday 4th December, an oral abstract session was held between 4:30pm and 6:00pm in the “Aggressive Lymphoma (Diffuse Large B-Cell and other aggressive B-Cell Non-Hodgkin Lymphomas) – results from prospective clinical trials: Novel Approaches in Aggressive Lymphoma” category. This session was moderated by Jason R. Westin, MD MS, of The University of Texas MD Anderson Cancer Center, and Laurie H Sehn, MD, of the British Columbia Cancer Agency.
Abstract #471 was presented during this session, titled “First Analysis of an International Double-Blind Randomized Phase III Study of Lenalidomide Maintenance in Elderly Patients with DLBCL Treated with R-CHOP in First Line, the REMARC Study from Lysa” by Catherine Thieblemont, MD, PhD, of Hospital Saint- Louis, Paris, France, and colleagues.
The group conducted the REMARC (NCT01122472) study, a multicenter, double-blind, randomized, placebo controlled phase III study, to determine the benefit of lenalidomide maintenance after response to R-CHOP in elderly patients with untreated DLBCL, FL3B or transformed lymphoma. Progression Free Survival (PFS) was the primary endpoint. Overall Survival (OS), safety, and Partial Response (PR) to Complete Response (CR) conversion were the secondary endpoints. Results obtained are:
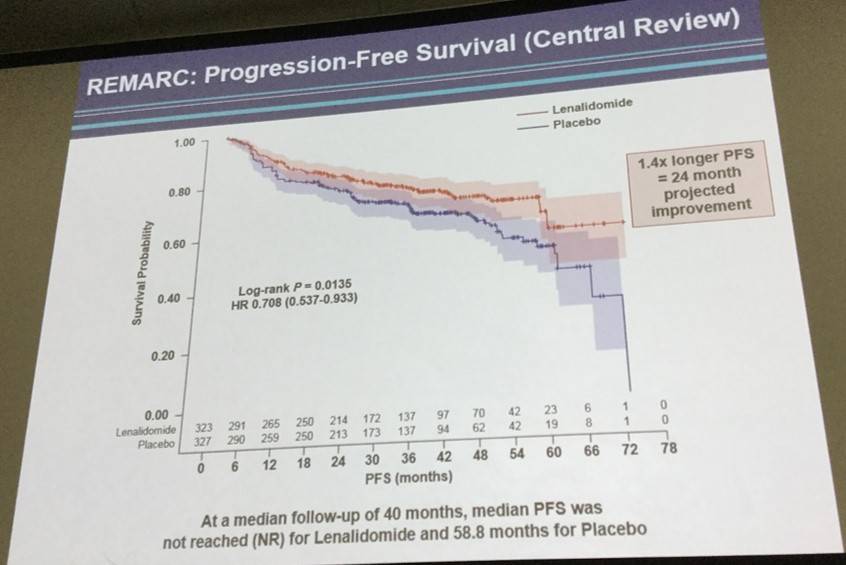
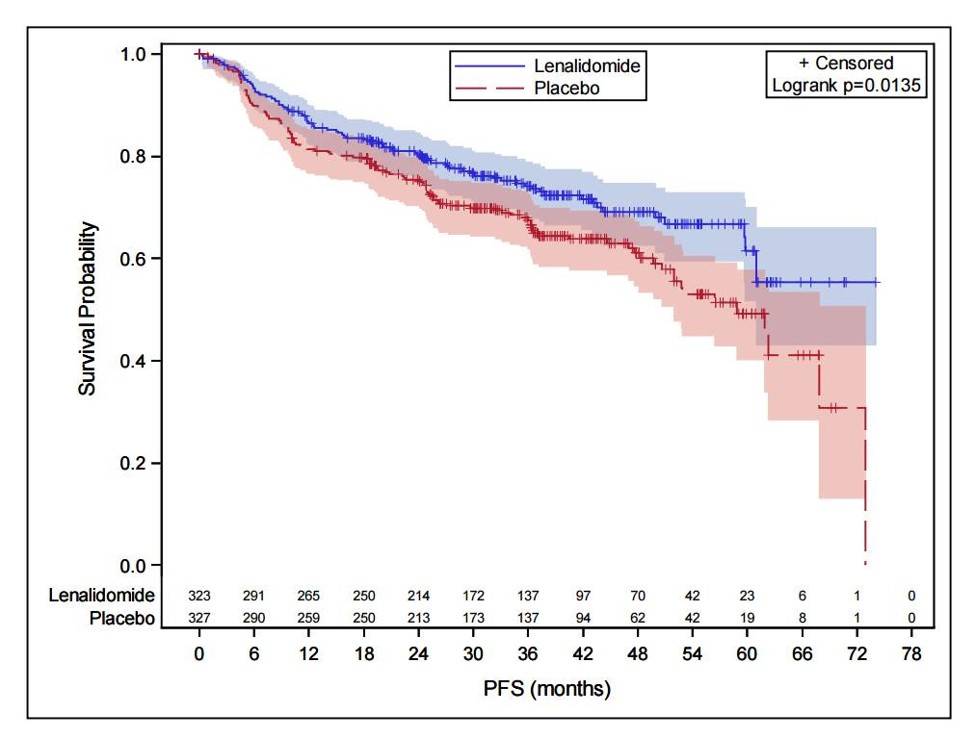
- Median follow-up of 40 months: median PFS was not reached in lenalidomide maintenance group vs. 58.8 months in the placebo (PBO) group. HR favored the lenalidomide group: 0.708 (95% CI: 0.537-0932; P = 0.0135)
- More patients converted from PR to CR during lenalidomide maintenance compared to PBO (18 vs 13)
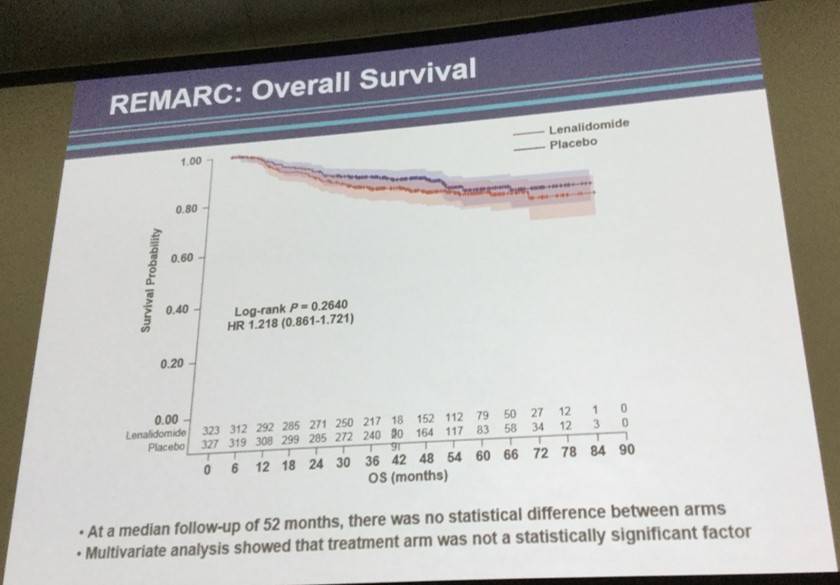
- No benefit for OS in the lenalidomide arm
- Deaths generally occurred off study drug: Median time from last dose of study drug to death was 277 days in the lenalidomide arm and 334 in control group
- Most frequent grade 3 or 4 AEs = neutropenia (56% vs 22%), rash (5% vs 1%), infections (8% vs 6%), thrombocytopenia (2.5% vs6%) in the lenalidomide and PBO arms, respectively
- Due to toxicity, patients stopped lenalidomide maintenance and PBO (59% vs 40%, P < 0.001)
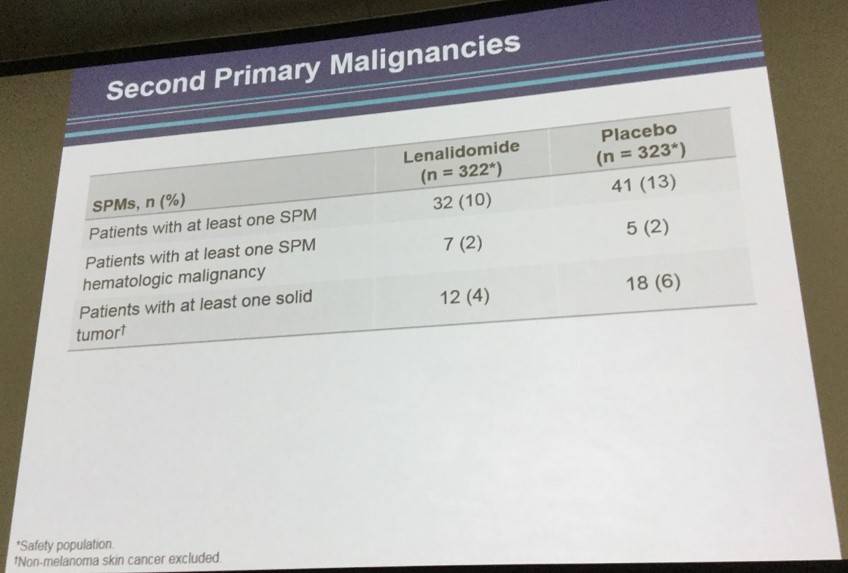
- Secondary primary malignancies occurred in 32 patients receiving lenalidomide and in 41 patients receiving PBO
The presentation was concluded by summarizing the REMARC study, which revealed that lenalidomide maintenance in DLBCL patients significantly improves the PFS without an early significant impact on OS. This study is the first report on the use of an immunomodulatory agent as maintenance therapy. The early data obtained shows that lenalidomide maintenance prolongs PFS for patients with DLBCL after treatment with R-CHOP. COO analyses are ongoing.

Abstract
Background. R-CHOP is the standard first-line treatment for elderly patients with diffuse large B-cell lymphoma (DLBCL). However 30% of patients will relapse and 70% of relapsed patients will die within 2 years of diagnosis. The REMARC study (clinicalTrials.gov NCT01122472) is an international, multicenter, double-blind, randomized, placebo controlled, phase III trial that assessed the benefit of lenalidomide (LEN) maintenance after response to R-CHOP in patients aged 60 to 80 years with untreated DLBCL, FL3b or transformed lymphoma. Patients achieving CR or PR at the end of 6 or 8 cycles of R-CHOP21 or R-CHOP14 were stratified by CR/PR status and country and randomized 1:1 to receive 2 years of LEN maintenance (25 mg/day for 21 of every 28 days) or placebo (PBO). The primary endpoint of the study was progression-free survival (PFS). Secondary endpoints were safety, PR to CR conversion rate, and overall survival (OS). Diagnosis was retrospectively centrally reviewed. In patients with adequate samples, GCB/nonGCB profile was assessed by the Hans algorithm and GCB/ABC/unclassified profile was assessed using NanoString gene expression profiling technology.
Methods. From 05/2009 to 05/2014, 784 patients were enrolled either before R-CHOP (n= 437) or after completion of 6 or 8 cycles of R-CHOP (n= 347). At the end of R-CHOP therapy, 650 patients were randomized to maintenance, either in CR (n= 495) or in PR (n= 152). Central review found that 3 patients were randomized in SD or PD, all in LEN arm. At time of diagnosis, median age was 68 y (range 58-80), 43.5% were older than 70 y, and 56% were male. aaIPI was low in 38.5% and high in 57.5% of patients (missing data 4%). COO analyses are ongoing for both Hans algorithm and NanoString technology.
Results. With a median follow-up of 40 months, median PFS (according to independent centralized radiology review) was not reached in the LEN group versus 68 months in the PBO group (hazard ratio favoring the LEN group, 0.708 (95% CI 0.537-0.932; p=0.0135))(See Figure). In the LEN group, 18 patients (21%) converted from PR to CR during maintenance compared to 13 patients (14%) in the PBO group. Immature overall survival data did not show any benefit for LEN arm, a lack of difference not attributable to an excess of lymphoma relapse, secondary cancer or safety problems in LEN arm. Deaths generally occurred off study drug (median time from last dose of study drug to death was 277 days (range 20, 1291) in LEN arm and 334 (41, 1594) in control arm. During maintenance, the most common observed grade 3 or 4 AEs were neutropenia (56% vs. 22%), rash (5% vs. 1%), infections (8% vs. 6%), and thrombocytopenia (2.5% vs. 0.6%) in LEN and PBO arms, respectively. Dose adjustments were necessary in 72% of the LEN patients and 42% of PBO patients. 59% of patients stopped LEN and 40% stopped PBO for toxicity (p<0.001). Median number of cycles was 15 in LEN and 25 in PBO (p<0.001). Secondary primary malignancies occurred in 33 patients receiving LEN and in 42 patients on PBO.
Conclusion. This analysis of the REMARC study shows that 2 years of LEN maintenance in patients responding to R-CHOP significantly improved PFS (primary endpoint) without an early significant impact on OS. The COO analysis is currently ongoing. This is the first report finding that using an immunomodulatory agent as maintenance therapy prolongs PFS for patients with DLBCL after first line treatment with R-CHOP.
Figure 1. Progression-free survival of elderly patients with diffuse large B-cell lymphoma in response to R-CHOP treated in maintenance with either lenalidomide or placebo
- Thieblemont C. et al. First Analysis of an International Double-Blind Randomized Phase III Study of Lenalidomide Maintenance in Elderly Patients with DLBCL Treated with R-CHOP in First Line, the REMARC Study from Lysa. 2016 December 4; Oral Abstract #471: ASH 58th Annual Meeting and Exposition, San Diego, CA.

Understanding your specialty helps us to deliver the most relevant and engaging content.
Please spare a moment to share yours.
Please select or type your specialty
 Thank you
Thank youRelated articles
Newsletter
Subscribe to get the best content related to lymphoma & CLL delivered to your inbox








