All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a Healthcare Professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The Lymphoma Hub uses cookies on this website. They help us give you the best online experience. By continuing to use our website without changing your cookie settings, you agree to our use of cookies in accordance with our updated Cookie Policy
Introducing

Now you can personalise
your Lymphoma Hub experience!
Bookmark content to read later
Select your specific areas of interest
View content recommended for you
Find out moreThe Lymphoma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Lymphoma Hub cannot guarantee the accuracy of translated content. The Lymphoma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
ASH 2016 | Results from phase II study of rituximab, bendamustine and cytarabine (RBAC500) as induction therapy in elderly patients with MCL
Bookmark this article
The 58th Annual Meeting & Exposition of the American Society of Hematology’s (ASH) took place in San Diego, CA, on December 3–6, 2016. On Sunday 4th December, an oral abstract session was held between 4:30pm and 6:00pm in the “Aggressive Lymphoma (Diffuse Large B-Cell and other aggressive B-Cell Non-Hodgkin Lymphomas) – results from prospective clinical trials: Novel Approaches in Aggressive Lymphoma” category. This session was moderated by Jason R. Westin, MD MS, of The University of Texas MD Anderson Cancer Center, and Laurie H Sehn, MD, of the British Columbia Cancer Agency.
Abstract #472 was presented during this session, titled “Rituximab, bendamustine and cytarabine (RBAC500) as induction therapy in elderly patients with Mantle Cell Lymphoma: Final Results of a phase 2 study from the Fondazione Italiana Linfomi” by Carlo Visco, MD, of the San Bortolo Hospital, Vincenza, Italy, and colleagues.
Hematologic toxicity, including thrombocytopenia and neutropenia, is relevant in patients with Mantle Cell Lymphoma (MCL) undergoing treatment with rituximab, bendamustine and cytarabine (R-BAC) 800mg/m2 (Visco et al., 2013). The group designed a phase 2 trial, RBAC500 (NCT01662050), aimed at reducing hematologic toxicity by adopting the R-BAC schedule, but lowering cytarabine to 500mg/m2 in previously untreated patients with MCL. Complete Remission (CR) measured by FDG-PET according to Cheson Criteria 2007 and safety were the primary endpoints. Molecular Response (MR), Progression Free Survival (PFS) and Overall Survival (OS) were the secondary endpoints. From May 2012 to February 2014, 57 patients (median age of 71 years [61-7]) were treated with a combination of R-BAC:
- Fifty-three patients received 4 cycles, thirty-six patients received 6 cycles
- Due to hemato-toxicity AEs between cycles, three patients discontinued treatment before cycle 4
- Grade 3 and 4 neutropenia and thrombocytopenia were observed in 49% and 52% of administered cycles, respectively
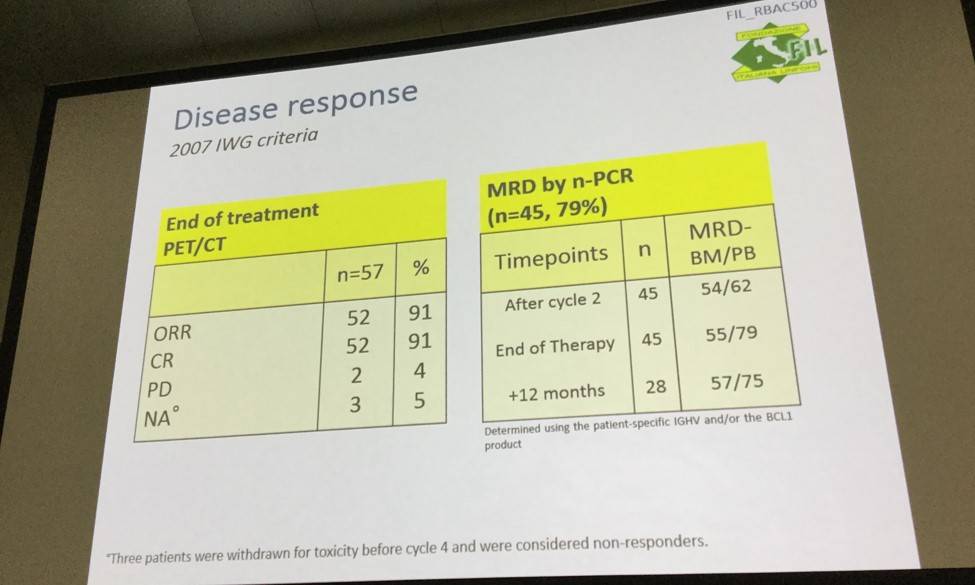
- Overall Response Rate (ORR) and CR were both 91%
- MRs were 79% and 55% in the peripheral blood and bone marrow samples, respectively
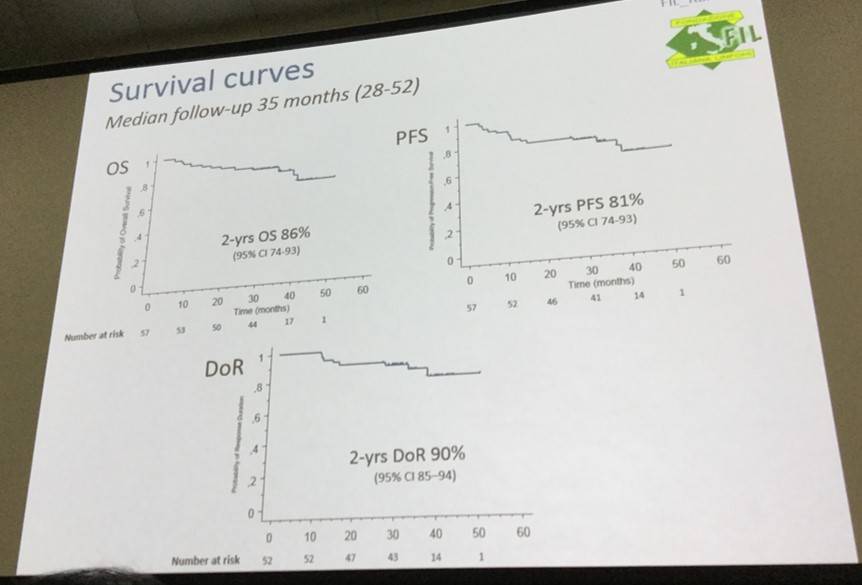
- At median follow up of 34 months, 2-year PFS and OS were 81% and 86%, respectively
- After 34 months, patients with elevated Ki-67 (>30%) and blastoid variant had significantly lower PFS compared to patients with classical/pleomorphic variants and low proliferative index (41% vs 97%; P = 0.0001) (Figure 1)
R-BAC500 regimen in patients with MCL was safely administered as first-line therapy and highly effective with 93% FDG-PET negative CR, and a 2-year PFS of 81% without maintenance therapy. During this oral presentation, it was concluded that hematologic toxicity is substantially reduced with the R-BAC500 regimen and it compares favorably with other regimens including R-bendamustine.

Abstract
Background: The combination of rituximab (R, 375 mg/m2 intravenously [IV], day 1), bendamustine (B, 70 mg/m2IV, days 2 and 3), and cytarabine (800 mg/m2, IV on days 2 to 4) was highly active in patients with mantle-cell lymphoma (MCL) in a phase 2 study [R-BAC; Visco et al, JCO 2013]. This regimen was well tolerated, but hematologic toxicity was quite relevant, especially in terms of transient grade 3 to 4 thrombocytopenia (76% of cycles). Aiming at reducing hematologic toxicity, the Fondazione Italiana Linfomi (FIL) designed a phase 2 trial adopting the R-BAC schedule, but lowering cytarabine dose to 500 mg/m2 (RBAC500).
Materials and Methods: Patients with newly diagnosed MCL, aged 61 to 80 years, not eligible for autologous transplant and fit according to the comprehensive geriatric assessment, were enrolled. Patients presenting with non-nodal leukemic disease were excluded. The primary endpoints were complete remission rate (CR) measured by FDG-PET according to Cheson criteria 2007, and safety. Secondary endpoints included rate of molecular response (MR) by nested-PCR using patient specific IGH or BCL1 based targets, progression-free (PFS) and overall survival (OS). The study was conducted according to the Bryant and Day two-stage design.
Results: From May 2012 to February 2014, 57 patients with MCL from 29 centers were recruited and treated. Central pathology revision was performed in 87% of cases. Median age was 71 years (range 61-79), 75% were males, and 91% had Ann Arbor stage III/IV disease. Mantle Cell International Prognostic Index (MIPI) was low in 15%, intermediate in 40%, high in 45%, Ki-67 was ≥30% in 31%, and 9% had the blastoid cytological variant. Overall, 53 patients (91%) received at least 4 cycles, while 36 (63%) had 6 cycles (median 5.3 cycles per patient). Fifteen patients (26%) discontinued treatment before reaching cycle 6 because of toxicity/adverse events, that mainly consisted of prolonged hemato-toxicity between cycles. Only one patient discontinued due to progressive disease. Grade 3 or 4 neutropenia and thrombocytopenia were observed in 49% and 52% of administered cycles, respectively. Febrile neutropenia occurred in 6% of cycles. Extra-hematologic toxicity was mainly cardiac (5%). Overall response rate was 96%, and CR was 93%. The MR rate at the end of treatment was 76% on peripheral blood and 55% on bone marrow (BM) samples. With a median follow-up of 34 months (28-52), the 2-years PFS (± confidence interval) was 81%±5% and the OS 85%±4%. Elevated Ki-67 (≥30%), and the blastoid variant were the strongest independent predictors of adverse PFS. Patients with either of these two features (33%), had a significantly inferior PFS (41% vs 97% after 34 months) compared to patients with classical/pleomorphic variants and low proliferative index (p<0.0001, Figure 1).
Conclusions: The R-BAC500 regimen can be safely administered as first line therapy to elderly patients with MCL. Hematologic toxicity is substantially reduced compared to our previous experience. With 93% of FDG-PET negative CR, and a 2-years PFS of 81% without maintenance therapy, the R-BAC500 regimen is a highly effective treatment for patients with MCL, and compares favourably with previously reported regimens in this patient population, including R-bendamustine. 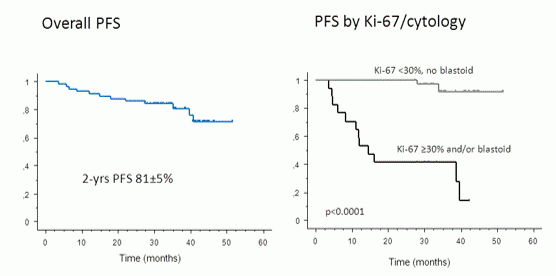
Figure 1: Progression-Free Survival Curve
- Visco C. et al. Rituximab, bendamustine and cytarabine (RBAC500) as induction therapy in elderly patients with Mantle Cell Lymphoma: Final Results of a phase 2 study from the Fondazione Italiana Linfomi. 2016 December 4; Oral Abstract #472: ASH 58th Annual Meeting and Exposition, San Diego, CA.
- Visco C et al. Combination of rituximab, bendamustine, and cytarabine for patients with mantle-cell non-Hodgkin lymphoma ineligible for intensive regimens or autologous transplantation. J Clin Oncol. 2013 Apr 10; 31(11):1442–1229. doi: 10.1200/JCO.2012.45.9842. Epub 2013 Feb 11.

Understanding your specialty helps us to deliver the most relevant and engaging content.
Please spare a moment to share yours.
Please select or type your specialty
 Thank you
Thank youRelated articles
Newsletter
Subscribe to get the best content related to lymphoma & CLL delivered to your inbox








