All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a Healthcare Professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The Lymphoma Hub uses cookies on this website. They help us give you the best online experience. By continuing to use our website without changing your cookie settings, you agree to our use of cookies in accordance with our updated Cookie Policy
Introducing

Now you can personalise
your Lymphoma Hub experience!
Bookmark content to read later
Select your specific areas of interest
View content recommended for you
Find out moreThe Lymphoma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Lymphoma Hub cannot guarantee the accuracy of translated content. The Lymphoma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
ASH 2016 | R2W: Subcutaneous Bortezomib, Cyclophosphamide and Rituximab (BCR) Versus Fludarabine, Cyclophosphamide and Rituximab (FCR) for Initial Therapy of Waldenström’s Macroglobulinemia: A Randomized Phase II Study
Bookmark this article
The 58th Annual Meeting & Exposition of the American Society of Hematology’s (ASH) took place in San Diego, CA, and on December 5th, Rebecca L. Auer, MD, PhD, from Barts Health NHS Trust, presented the results of a randomized, multi-center phase-II study assessing the efficacy and safety of the addition of cyclophosphoamide to the combinations of bortezomib + rituximab or fludarabine + rituximab in untreated Waldenström’s Macroglobulinemia (WM) patients.
Highlights:
- BCR= Six 28 day cycles of 1.6mg/m2 Bortezomib (SC) and 250mg/m2 Cyclophosphoamide (oral) on days 1, 8 and 15 plus 375mg/m2 Rituximab (IV) on days 1, 8, 15, 22 in cycles 2 and 5
- FCR= Six 28 day cycles of 40mg/m2 Fludarabine (oral) and 250mg/m2 Cyclophosphoamide (oral) on days 1-3 plus 375mg/m2 Rituximab (IV) on days 1, 8, 15, 22 in cycles 2 and 5
- 59 ITT patients, BCR:FCR 2:1, BCR n=42, FCR n=17
- Six cycles completed by: BCR= 92.9% with 38.1% pts having dose reductions, FCR= 76.5% with 52.9% pts having dose reductions
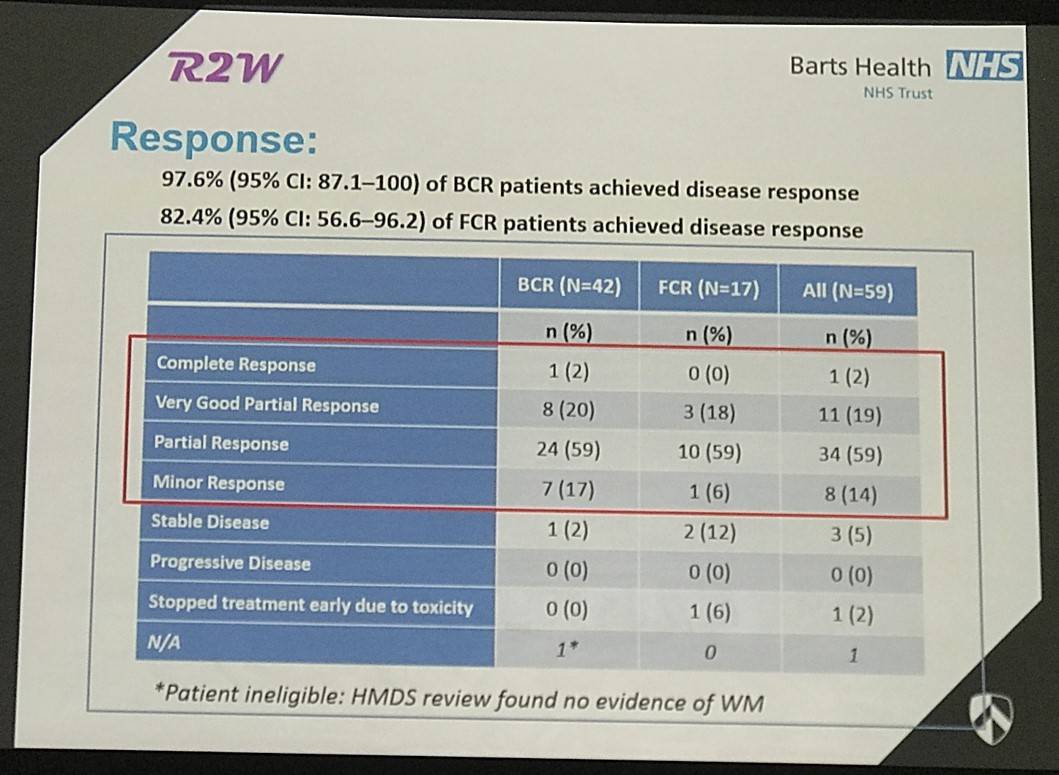
- ORR: FCR= 82.4%, BCR=97.6%
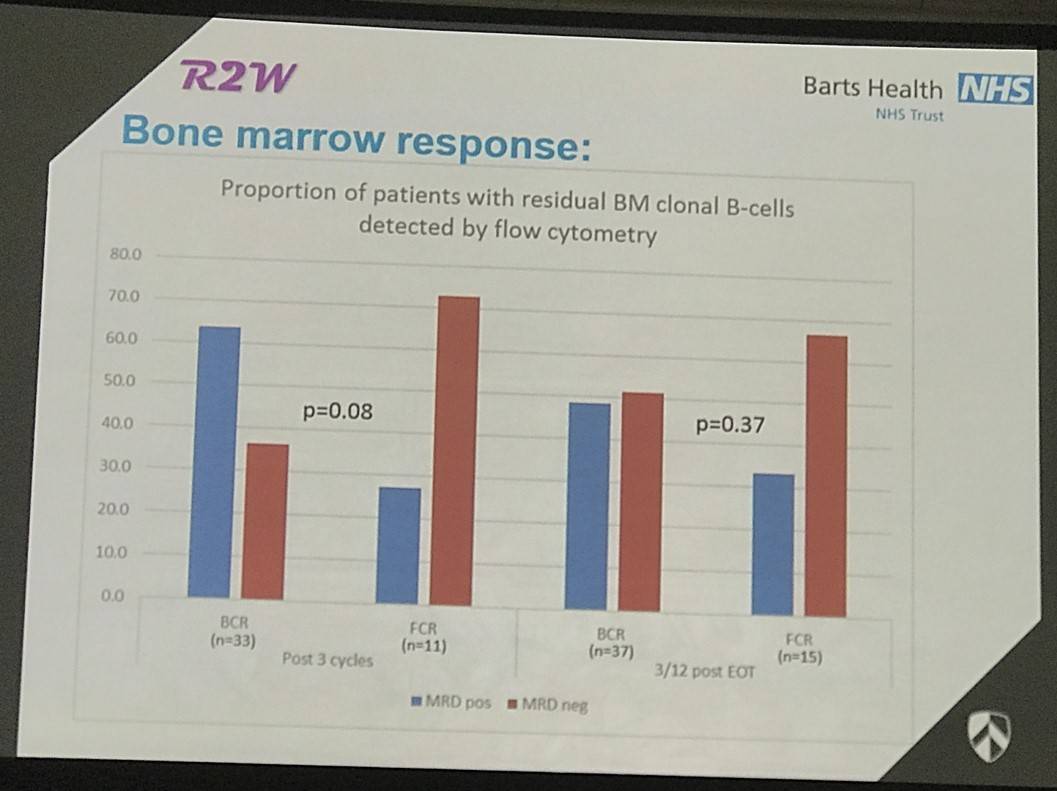
- At 18months follow-up 3 patients had died, 2 of which from myelodysplastic syndrome MDS who had received FCR
- FCR was seen to consistently have higher rates of ≥grade 3 toxicities than BCR
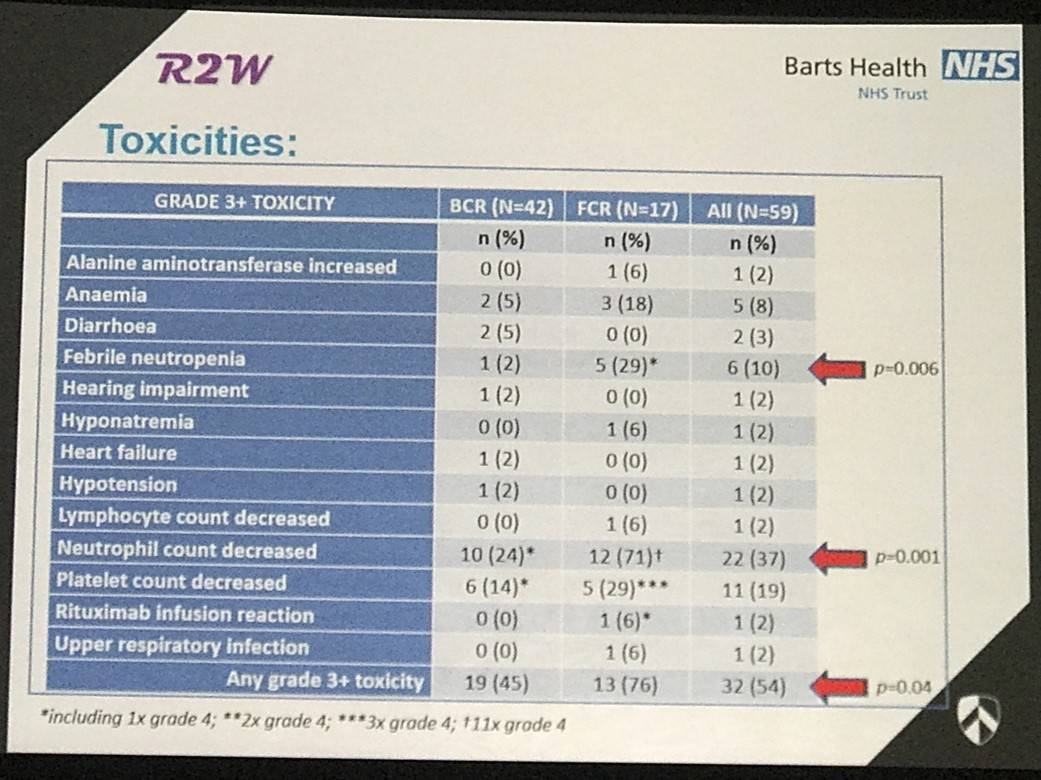
Rebecca Auer concluded by summarizing that FCR is associated with higher levels of toxicity and MDS than BCR, however both were effective treatments. Follow-up for this trial is ongoing, but BCR study in a Phase-III trial in WM was deemed to be warranted.

Abstract:
Introduction: Earlier studies have indicated that the combination of bortezomib and rituximab is highly active in Waldenstrőm’s macroglobulinemia (WM). However, there is scope to improve the complete response rate, duration of response and toxicity profile. We evaluated the efficacy and tolerability of the addition of cyclophosphamide to bortezomib and rituximab in previously untreated patients with WM.
Methods: Symptomatic treatment-naïve patients were enrolled into this prospective randomised (2:1), multicentre, non-comparative Phase II study (NCT01592981). Patients were stratified according to the International Prognostic Scoring System for WM. Patients were treated with BCR (Bortezomib 1.6 mg/m2 s.c. days 1, 8, 15; Cyclophosphamide 250 mg/m2 oral days 1, 8, 15; Rituximab 375mg/m2 i.v. days 1, 8, 15, 22 cycles 2 and 5 only) or FCR (Fludarabine 40mg/m2 oral days 1-3; Cyclophosphamide 250 mg/m2 oral days 1-3; Rituximab 375mg/m2i.v. days 1, 8, 15, 22 cycles 2 and 5 only) for 6 cycles repeated every 28 days. Rituximab and bortezomib were provided free of charge by Roche and Janssen, respectively. The primary endpoint was investigator assessed overall response rate (ORR) using consensus criteria.
Results: Sixty patients were enrolled into this study and 59 received trial treatment (BCR=42, FCR=17). Of all registered patients, 73% were male, median (range) age was 67 years (43-87), Haemoglobin 9.8 g/dL (6.5-14.0), serum IgM paraprotein 34 g/L (3.2-80.2), plasma viscosity 3.6 mPa.s (2.0-9.3) and 25/30/45% were low/intermediate/high risk respectively. Six cycles were completed by 92.9% of BCR and 76.5% of FCR patients, one patient withdrew from the study prior to starting trial treatment. Dose reductions were needed in 38.1% of BCR and 52.9% of FCR patients and treatment delays occurred in 64.3% of BCR and 64.7% of FCR patients. ORR was 97.6% in BCR patients with 78.6% achieving a major response (CR=1, VGPR=8, PR=24, MR=7, SD=1), one patient was not assessed as no evidence of WM was found upon central review; 82.4% in FCR patients with a major response rate of 76.5% (CR=0, VGPR=3, PR=10, MR=1, SD=2), one patient stopped treatment after cycle 1 due to continuing cytopenia (grade 4). Responses were also evaluated in both marrow and peripheral blood using a disease specific multiparamater flow cytometric assay. After a median follow-up of 18 months, 54 patients were progression-free; 3 patients progressed (all BCR) and 3 patients died, 2 from myelodysplastic syndrome (MDS) (both FCR) and 1 from pneumonia (BCR). Grade 3 or higher toxicities included anemia (5 [11.9%] BCR; 3 [17.6%] FCR), neutropenia (11 [26.2%] BCR; 12 [70.6%] FCR), thrombocytopenia (7 [16.7%] BCR; 6 [35.3%] FCR) and infection (2 [4.8%] BCR; 5 [29.4%] FCR). No grade 3 or higher neuropathy was reported.
Conclusions: BCR and FCR are both highly effective treatments for primary therapy of WM but FCR is associated with increased toxicity and concerning incidence of secondary MDS. BCR warrants further investigation in a randomised Phase III trial. Continued follow-up of R2W patients is also important to provide reliable estimates for duration of response, progression-free survival and overall survival.
- Auer R. et al. R2W: Subcutaneous Bortezomib, Cyclophosphamide and Rituximab (BCR) Versus Fludarabine, Cyclophosphamide and Rituximab (FCR) for Initial Therapy of Waldenstrőm’s Macroglobulinemia: A Randomised Phase II Study. 2016 December 5; Oral Abstract #618: ASH 58th Annual Meeting and Exposition, San Diego, CA.

Understanding your specialty helps us to deliver the most relevant and engaging content.
Please spare a moment to share yours.
Please select or type your specialty
 Thank you
Thank youRelated articles
Newsletter
Subscribe to get the best content related to lymphoma & CLL delivered to your inbox








