All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a Healthcare Professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The Lymphoma Hub uses cookies on this website. They help us give you the best online experience. By continuing to use our website without changing your cookie settings, you agree to our use of cookies in accordance with our updated Cookie Policy
Introducing

Now you can personalise
your Lymphoma Hub experience!
Bookmark content to read later
Select your specific areas of interest
View content recommended for you
Find out moreThe Lymphoma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Lymphoma Hub cannot guarantee the accuracy of translated content. The Lymphoma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
ASH 2016 | Outcome of Patients with Primary Refractory or Relapsed Peripheral T-Cell Lymphoma
Bookmark this article
The 58th Annual Meeting & Exposition of the American Society of Hematology (ASH) took place in San Diego, CA, on December 3–6, 2016.
On Monday 5th December, an oral abstract session was held between 2.45pm and 4.15pm in the “Hodgkin Lymphoma and T/NK Cell Lymphoma – Clinical studies: prognosis and effects of treatment in Hodgkin Lymphoma and Peripheral T-Cell Lymphomas” category. This session was moderated by Kara Kelly, MD, from the Roswell Park Cancer Institute, and Massimo Federico, MD, from the Università di Modena e Reggio Emilia.
Abstract #921 was presented by the co-moderator of this session, Massimo Federico, MD, and colleagues titled “The Outcome of Patients with Primary Refractory or Relapsed Peripheral T-Cell Lymphoma: Analysis of 1020 Cases Registered in the Prospective T-Cell Project”.
Massimo Federico and his group carried out an analysis on data obtained from the prospective registry T-cell project (NCT01142674), which analyzed the clinical features and factors influencing the survival of patients with Relapsed (Rel) or Refractory (Ref) (R/R) Peripheral T-Cell Lymphoma (PTCL). The key highlights are:
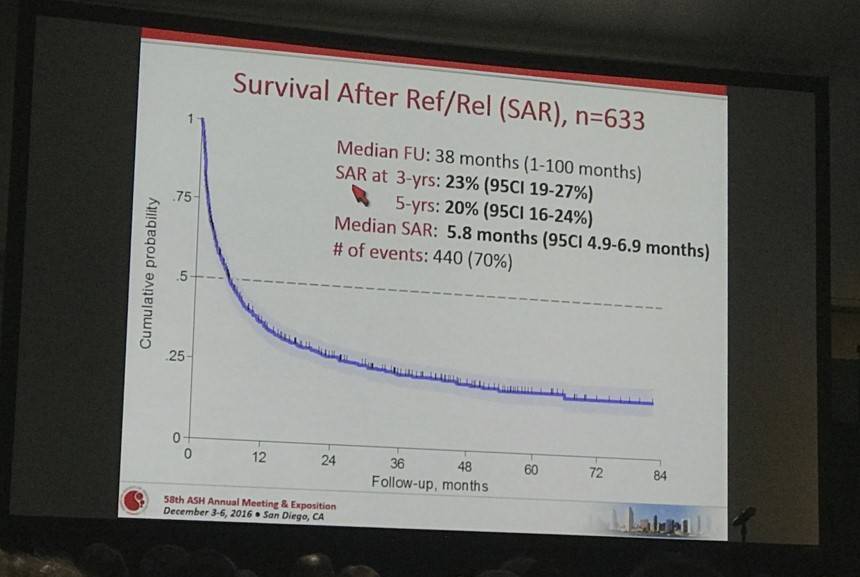
- At median follow up of 38 months, median survival after R/R (SAR) was 5.8 months
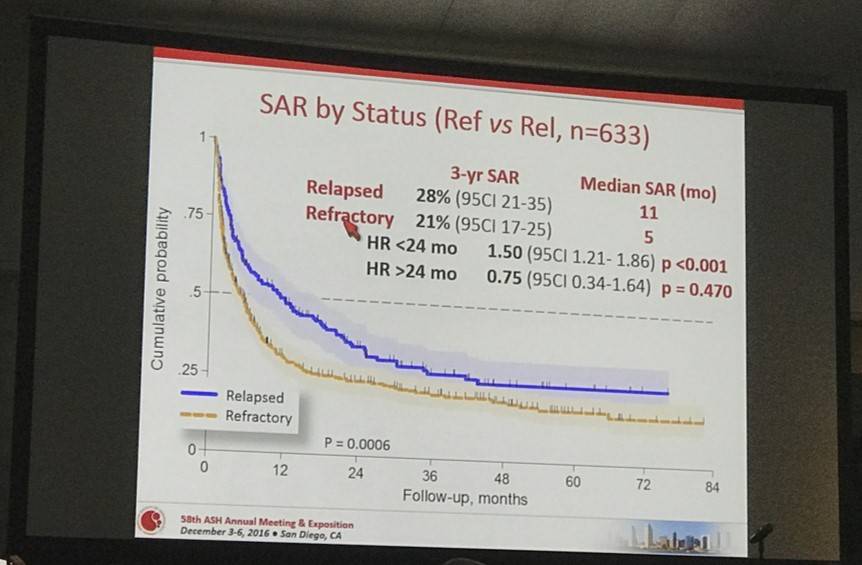
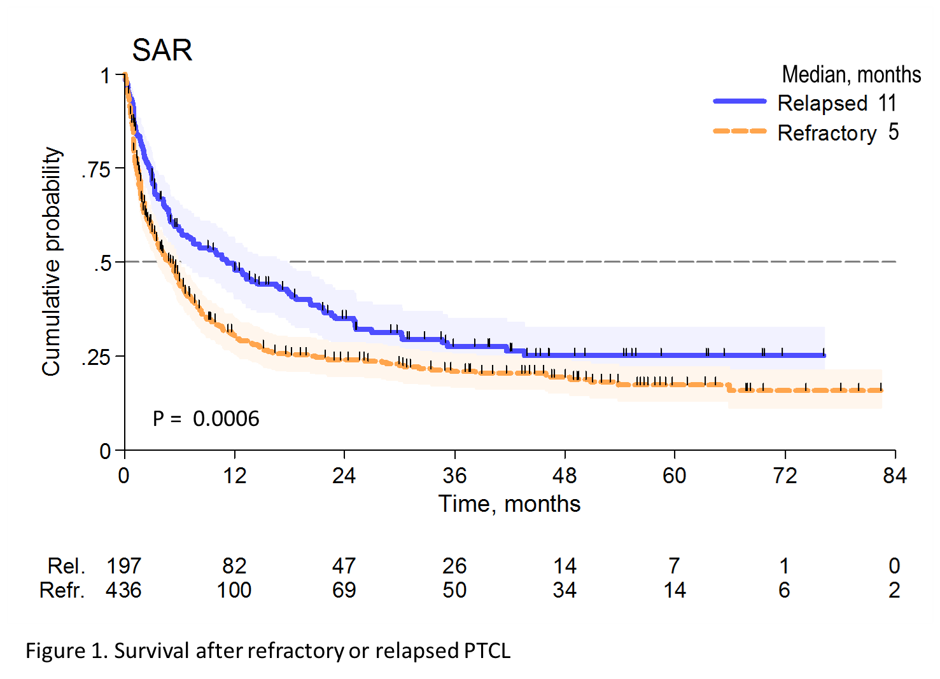
- 3-year SAR for Rel vs Ref; 28% vs 21%; HR < 24 months = 1.50 (P < 0.001)
- 3-year SAR for patients responding to salvage therapy consolidated with Autologous Stem Cell Transplantation (ASCT) was 48% compared to patients treated with high dose therapy; HR = 0.36, P = 0.001
- Higher risk of death was associated with Ref disease; HR = 2.14, P < 0.001
- Late Rel (> 12 months) had a better outcome; HR = 0.54, P < 0.001
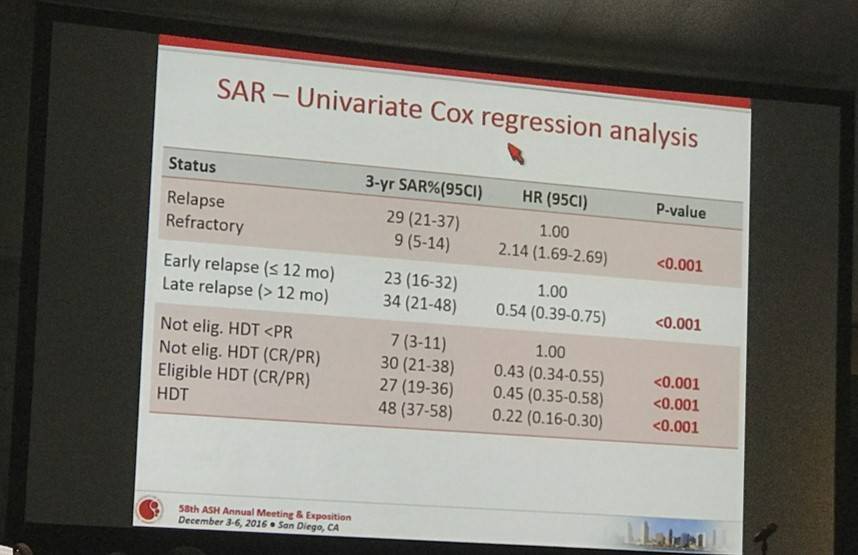
- Salvage therapy with ASCT also had a better outcome; HR = 0.35, P < 0.001
Massimo Federico concluded this oral presentation by highlighting that the SAR of PTCL patients with R/R remains dismal. Patients with Ref disease have a worse outcome than patients with Rel disease. The results obtained from this analysis suggests the need for novel agents and also effective salvage therapies for treatment of PTCL. Ongoing trials are currently underway in R/R PTCL patients using a combination of novel agents with chemotherapy with an aim to improve overall response in front line therapy.

Abstract
Introduction: Current treatment strategies for mature or peripheral T-cell lymphomas (PTCL) patients (pts) are largely unsatisfactory. In particular, pts failing first line therapy are expected to have a dismal outcome. The purpose of the present analysis was to analyze clinical features and explore factors influencing survival of pts with primary refractory or relapsed PTCL retrieved from a prospective registry, the T-cell Project.
Methods: In this study, data were extracted from cases of newly diagnosed PTCL registered by 74 institutions world-wide in the T-cell Project and on whom baseline data, information on first line treatment, response to initial therapy, time to relapse and salvage treatment were available. For this analysis, refractory disease (Ref) was defined as no response to initial treatment or unsatisfactory partial remission (PR) requiring salvage therapy immediately after completing frontline treatment. Relapsed disease (Rel) was defined as progression at least one month from completion of front line therapy in pts who achieved a complete remission (CR) or PR.
Results: Of the 1020 cases reviewed, 83 (8%) received palliative treatment and were excluded from this analysis. Out of 937 who received an active treatment (92%), 436 were classified as Ref (47%) and 197 as Rel (21%). Median time to relapse was 8 months (range 2-73 months). Overall, 77%, 73%, 59% and 45% of PTCL-NOS, AITL, ALCL alk- and ALCL alk+ were Rel/Ref, respectively. Forty-four percent of the pts were at high-risk according to PIT and 45% according to IPI. All but 3 pts received combination chemotherapy and 34 (5%) were consolidated with high dose therapy (HDT) in first remission. Additionally, 99 (16%) pts received HDT as part of salvage treatment. After a median follow-up from documentation of Rel/Ref disease of 38 months (range 1-96 months), 440 patients had died. The median survival after Rel/Ref disease (SAR) was 5.8 months (95% CI 4.9-7.2 months). At 3 years, the SAR was 28% for Rel (95% CI 21-35%) and 21% (95% CI 17-25%) for Ref (p<0.001) (Figure 1). Patients responding to salvage therapy and consolidated with autologous stem cell transplantation (ASCT) had a 3 year SAR of 48% (95% CI 37-58) with an HR of 0.36 (95% CI 0.26-0.48) compared to pts not treated with HDT (p<0.001). In a univariate Cox regression analysis, Ref disease was associated with a higher risk of death (HR=2.14, 1.69-2.69, p<0.001), whereas late relapse (>12 months, HR 0.54, 95% CI 0.39-0.75, p<0.001) and salvage therapy with ASCT (HR=0.35, 95% CI 0.26-0.48, p<0.001) were associated with a better outcome. No difference was found in outcomes for Rel/Ref pts with respect to histology.
Conclusion: The T-cell Project represents the largest cohort of prospectively collected data on pts with aggressive T-cell lymphomas and accurately reflects outcomes for pts treated according to standards of care around the world. We demonstrate that the SAR of pts with Rel/Ref PTCL remains dismal. Rel pts had a better outcome than those with primary Ref disease. While ASCT appeared to be beneficial for Rel/Ref pts, only a small percentage of pts were able to undergo transplant. These results highlight the urgent need for novel agents and more effective salvage therapies. Clinical trials are underway exploring the activity of novel agents in combination with chemotherapy to improve overall response in the front line, and single agent and combination studies of novel agents are underway for patients with Rel/Ref disease.
- Bellei M. et al. The Outcome of Patients with Primary Refractory or Relapsed Peripheral T-Cell Lymphoma: Analysis of 1020 Cases Registered in the Prospective T-Cell Project. 2016 December 5; Oral Abstract #921: ASH 58th Annual Meeting and Exposition, San Diego, CA.

Understanding your specialty helps us to deliver the most relevant and engaging content.
Please spare a moment to share yours.
Please select or type your specialty
 Thank you
Thank youRelated articles
Newsletter
Subscribe to get the best content related to lymphoma & CLL delivered to your inbox








