All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a Healthcare Professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The Lymphoma Hub uses cookies on this website. They help us give you the best online experience. By continuing to use our website without changing your cookie settings, you agree to our use of cookies in accordance with our updated Cookie Policy
Introducing

Now you can personalise
your Lymphoma Hub experience!
Bookmark content to read later
Select your specific areas of interest
View content recommended for you
Find out moreThe Lymphoma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Lymphoma Hub cannot guarantee the accuracy of translated content. The Lymphoma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
ESMO 2016 | Educational Session – EATCL: Enteropathy associated T-cell lymphomas
Bookmark this article
Dr Otto Visser from the Department of Hematology, VU University Medical Center, Amsterdam, presented a lecture during an educational session at this year’s ESMO annual meeting, held in Copenhagen, Denmark, on 7–11 October. His talk was titled: “Enteropathy associated with T-cell lymphomas.”
The talk began with a few slides that gave a brief overview describing coeliac disease, its symptoms, and the spectrum and progression of the diseases.

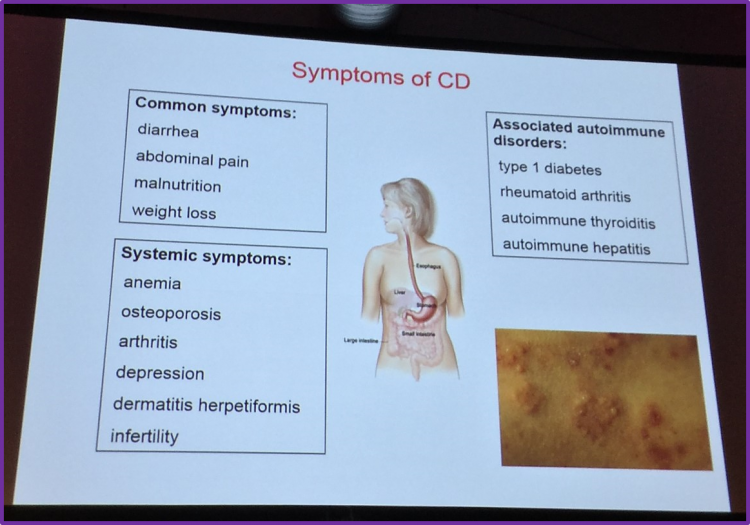

Following this brief overview, Dr Visser then described Refractory Coeliac Disease (RCD) in more detail, explaining that it is persistent or recurrent malabsorptive symptoms and villous atrophy, despite strict adherence to a Gluten-Free Diet (GFD) for at least 6–12 months and the absence of other causes of villous atrophy.
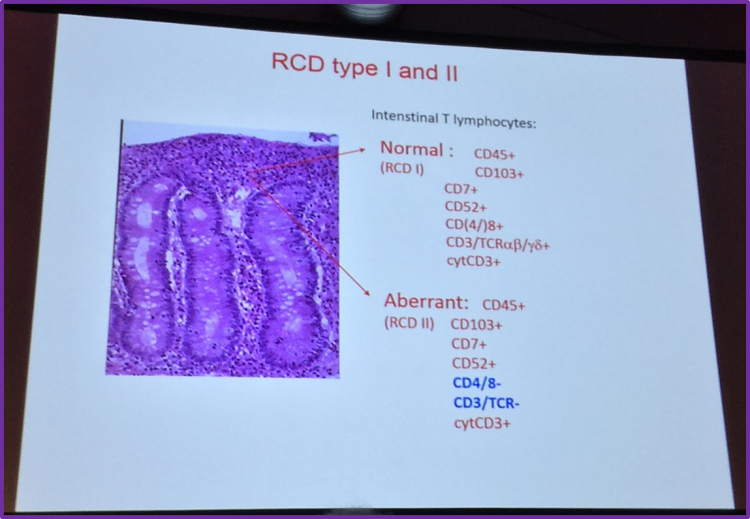
He then went on to explain that there are two types of RCD, which are distinguished by either the absence or presence of aberrant Intraepithelial Lymphocytes (IEL). In RCD I, there are no aberrant cells. Normal expression of T-cell antigens and oligo-/polyclonal TCR-γ gene rearrangement is observed. RCD II is identified when an aberrant IEL population is present (>20%) and in the majority of cases monoclonal TCR-γ gene rearrangement is observed.
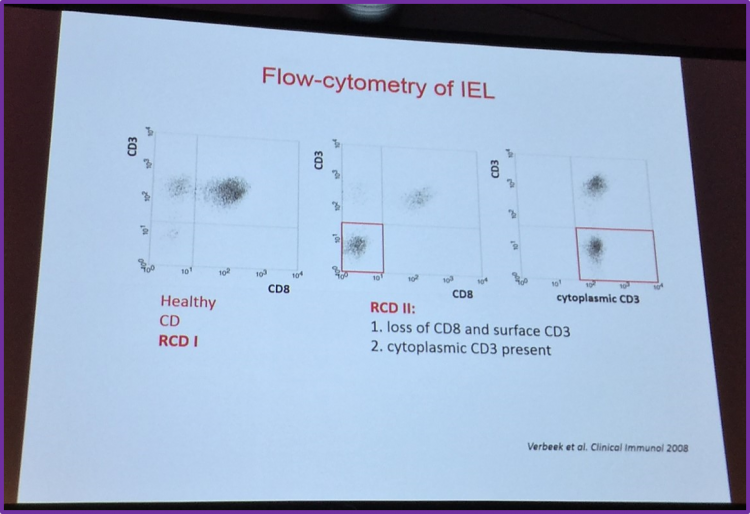
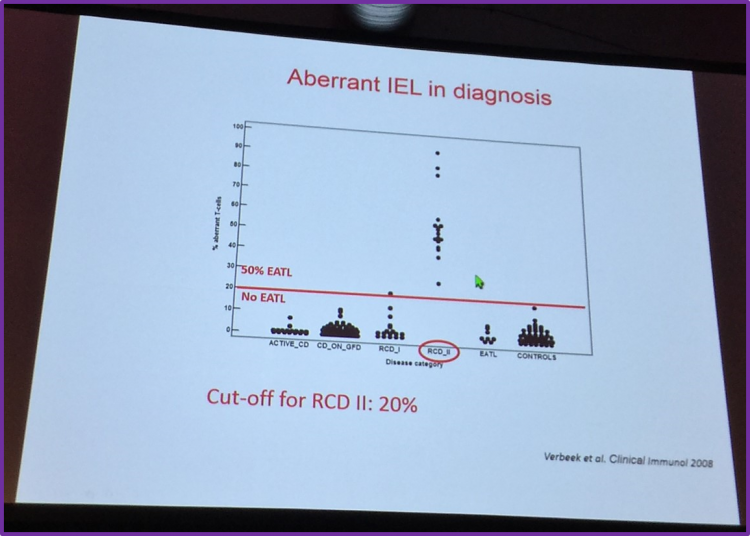
Visser also shared flow cytometry data published by Verbeek et al. in Clinical Immunology in 2008 which found that IELs in RCD II do not express surface CD3 and CD8 but do contain cytoplasmic CD3. Verbeek et al. found that quantifying aberrant IELs by flow cytometry is superior to T-cell clonality analysis for identifying patients with RCD at risk of EATL development. They also found that, when using flow cytometry, if more than 20% of IELs showed aberrancy then this resulted in a much higher negative predictive value and sensitivity (both 100%) for EATL development than T-cell clonality analysis (75% and 78%, respectively). Verbeek et al. concluded that a cut-off value of 20% is of use in risk stratification, informing future therapy and follow-up of patients with RCD.
The talk then focused on treatment for RCD types I and II:
A retrospective study comparing the medical files of 14 RCD I patients and 43 RCD II patients byMalamut et al. was published in Gastroenterology in 2009, which found that the presentation and prognosis of RCD II is much more severe than RCD I; the 5-year survival rates in RCD I and RCD II patients were 93% and <44%, respectively. They also conclude that aberrant IELs are a predictive factor but not a necessary requirement for the development of lymphoma.
Goerres et al. published their findings in Alimentary Pharmacology and Therapeutics in 2003 who assessed azathioprine and prednisone combination therapy for the treatment of RCD. In RCD I patients, 80% (8/10) responded histologically, and in four patients complete villi normalization was observed. However, azathioprine and prednisone were not effective for the treatment of RCD II; 6/8 patients developed EATL and 7/8 patients died.
Al-Toma et al. performed retrospective evaluation which aimed to provide insight into RCD and EATL development by reporting on risk of transition and long-term survival in a large patient cohort with complicated CD. Their findings were published in Gut in 2007. They found that, during the mean follow-up time of 5 years (range 2–15 years), no patient with RCD I treated with azathioprine and prednisone developed RCD II or EATL. However, over half (52%) of RCD II patients developed EATL within 4–6 years of RCD II diagnosis. The group concluded that more targeted and aggressive treatments are required for RCD II and EATL.
Dr Visser also referenced a review article by Woodward who published in Clinical and Experimental Gastroenterology in 2016, and a single center experience published by Rubio-Tapiaet al. which proposes a staging model which they hypothesize may improve the precision of prognosis in RCD patients (Gastroenterology, 2009).

Due to its dismal prognosis and unresponsiveness to current treatment strategies, the feasibility, safety and efficacy of Autologous Stem Cell Transplantation (ASCT) has been investigated to treat RCD II. Al-Toma et al., who published in Blood in 2007, performed ASCT (preceded by fludarabine and melphalan conditioning) in seven patients with RCD II. Engraftment was successful in all patients, no major non-hematologic toxicity or transplantation mortality occurred, and significant reduction in aberrant IELs were reported resulting in improvement in clinical well-being and normalization of hematologic biochemical markers. 8 months after transplantation, one patient died from progressive neurocoeliac disease. Al-Toma et al. conclude that their preliminary results showed that ASCT preceded by high-dose chemotherapy appears to have an acceptable safety profile and may present a new treatment option in patients with RCD II who do not respond to currently available drugs.
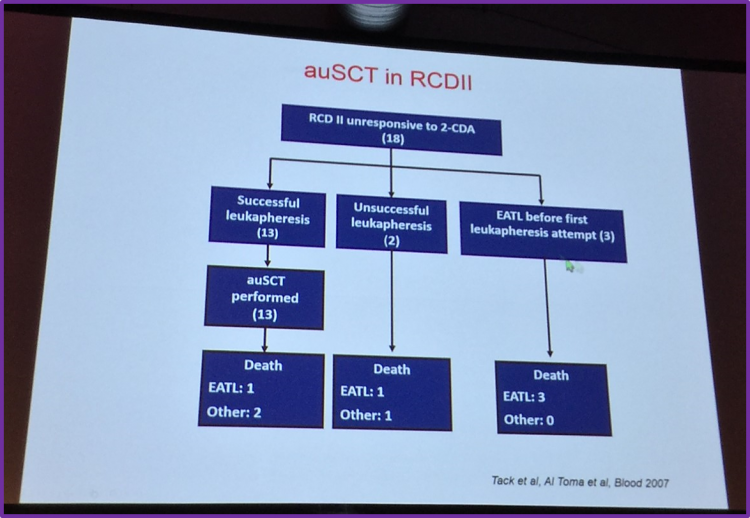
More recently, in 2011, Tack et al. published data in Bone Marrow Transplantation concerning ASCT in RCD II patients who were unresponsive to treatment with cladribine. From March 2004 to February 2010, 18 patients with RCD II were evaluated for conditioning with fludarabine and melphalan, followed by ASCT. Thirteen patients were successfully transplanted and a 4-year survival rate of 66% was reported. Most patients showed clinical improvement and 5 achieved a complete histological remission. ASCT could not be performed in five patients, they all died with a median survival of 5.5 months. After 4 months of follow-up, progression to EATL was observed in one transplanted patient. Tack et al. conclude that high-dose chemotherapy followed by ASCT is feasible and appears promising in the treatment of patients with RCD II who are unresponsive to cladribine.
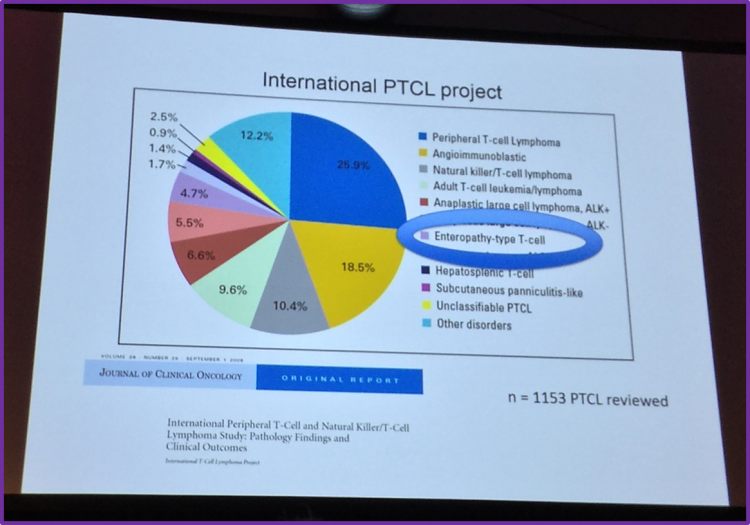
Dr Visser then moved on to discuss the International Peripheral T-Cell (PTCL) and Natural Killer/T-Cell Lymphoma (NKTCL) Study, published by Vose et al. in the Journal of Clinical Oncology in 2008. Of 1,153 confirmed cases of PTCL or NKTCL, enteropathy-type PTCL represented 4.7% of cases. It was also found that enteropathy-type PTCL has a 5-year Overall Survival (OS) of 20%. 5-year Failure-Free Survival (FFS) of enteropathy-type PTCL was 4%. Vose et al. concluded that the frequency of enteropathy-type PTCL in Europe is increasing; there is a known association between gluten-sensitivity and enteropathy, partly due to the higher frequency of Human Leukocyte Antigen (HLA)-DQ2 and HLA-DQ8 alleles in the northern European population. They also concluded that enteropathy-type PTCL was among the histologic subtypes with the worst outcomes and for which IPI was not helpful.
Following this, Dr Visser then summarized the criteria and findings of a paper published in Bloodin 2011 by Delabie et al. which reported clinical and histological findings specifically on EATL from the international PTCL project. They found that CHOP/CHOP-like regimens without high-dose chemotherapy and ASCT resulted in a Complete Response (CR) of 40% and a 5-year OS of 20% in patients with EATL.

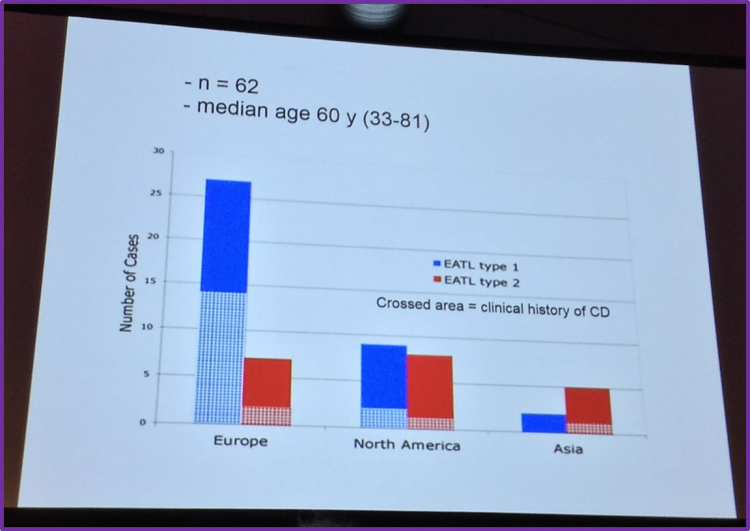
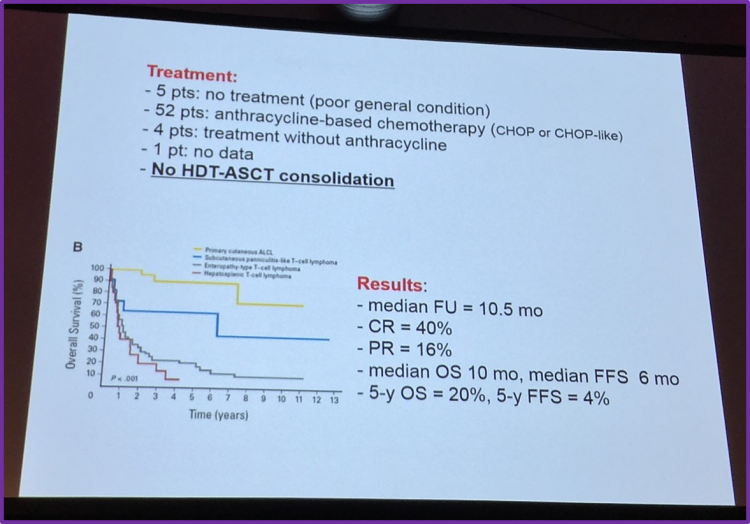
Pts, Patients; HDT, High-Dose Therapy; ASCT, Autologous Stem Cell Transplant; FU, Follow-Up; CR, Complete Response; PR, Partial Response; OS, Overall Survival; FFS, Failure-Free Survival
Dr Visser also summarized findings published in Blood in 2010 by Sieniawski et al.; from 1979 onwards, the Scotland and Newcastle Lymphoma Group (SNLG) prospectively collected data on all newly diagnosed patients with lymphoma living in Scotland or the North of England. Their findings indicated that the IVE-MTX-ASCT (ifosfamide, etoposide, epirubicin/methotrexate) treatment strategy has an acceptable toxicity profile and significantly improved outcome; the 5-year OS and PFS were both 68% for patients who completed the whole treatment, and 50% and 33% for patients who did not. The difference was statistically significant for PFS (P = .028) but not for OS (P = .251). Dr Visser also drew attention to the comparison made in this study between chemotherapy with or without surgery compared to just surgery alone.

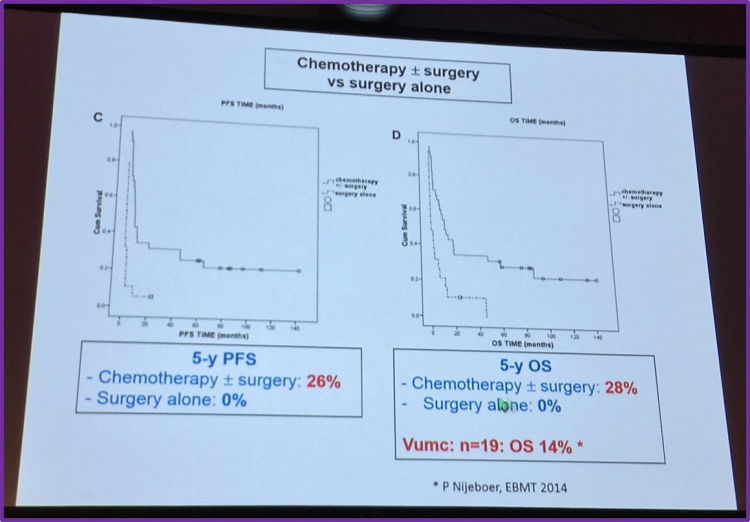
The speaker summarized this portion of the talk by stating that ASCT appears feasible in over half of patients and evidence has been found that ASCT can provide long-term remission. However, he asked if IVE-MTX is much better than CHOP before ASCT?
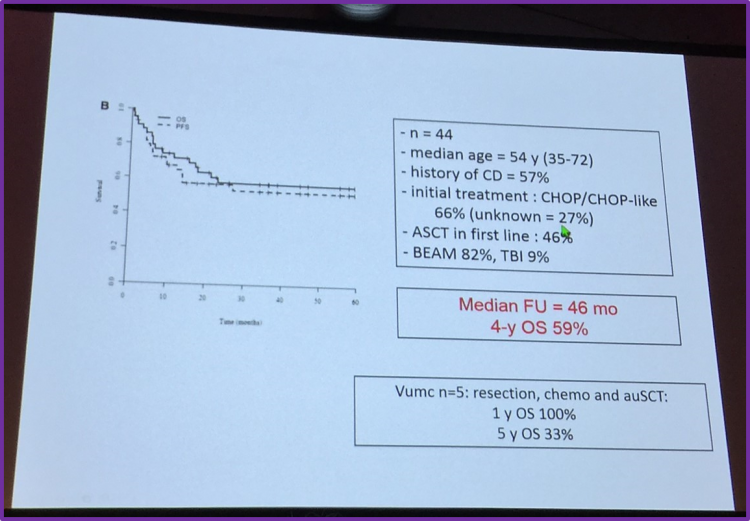
To answer this, Dr Visser summarized a retrospective study performed by the EBMT, published inBlood in 2013 by Jantunen et al. He drew particular attention to the fact that ASCT was found to provide long-term remission (4-year OS of 59%).

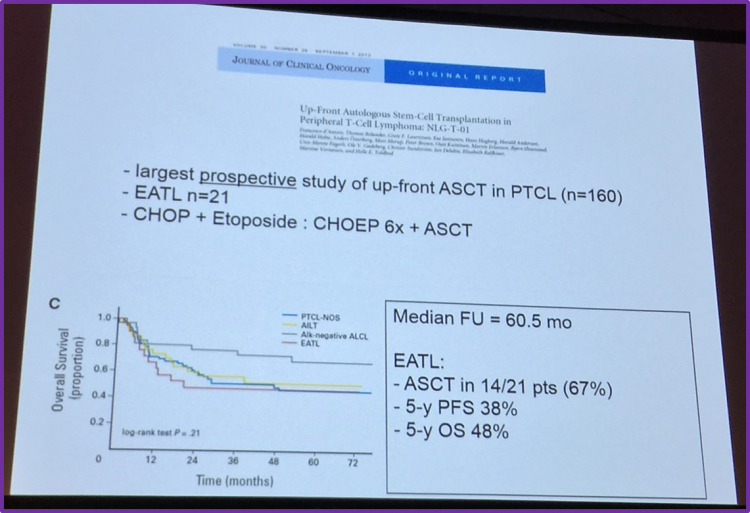
Moreover, Dr Visser also summarized findings published by D’Amore et al. in the Journal of Clinical Oncology in 2012. A total of 166 patients, aged 18–67, with newly diagnosed PTCL were enrolled between October 2001 and October 2007. 24 centers took part in this clinical trial by the Nordic Lymphoma Group. Dr Visser made a particular note of the finding that ASCT was feasible in 67% of included patients.
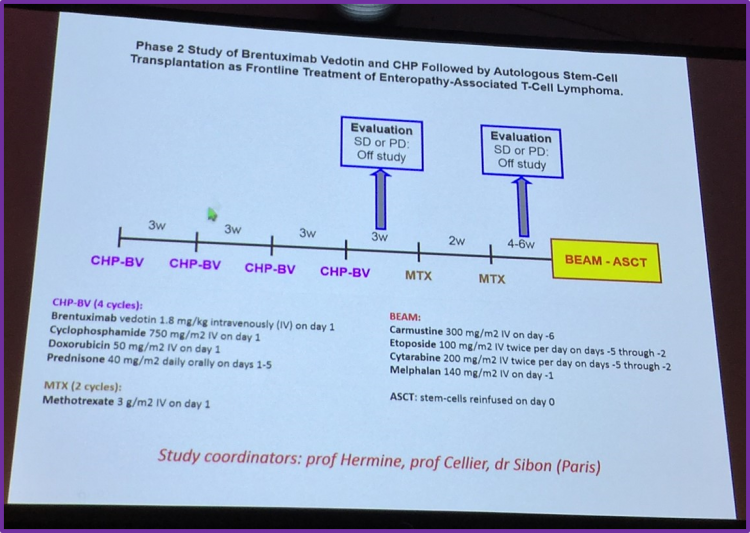
Dr Visser concluded this part of his talk by iterating that there is no consensus for initial treatment for EATL. Surgery should be considered as first-line treatment, but never alone. Chemotherapy should be considered whenever feasible but after CHOP without ASCT the CR rate is only 40% and the 5-year OS is only 20%. He once again stated that ASCT is feasible and can provide long-term remission. However, only 60% of patients who have an ASCT planned are actually transplanted. Therefore, treatment for EATL needs improving. New drugs and new combinations are needed to increase the response rate, increase the number of transplanted patients while being less toxic. Anti-CD30 brentuximab vedotin is potentially a good candidate. This is due to the fact that 90% of RCD I and II cases have been found to be CD30 negative and CD30 is rarely expressed by aberrant IELs. However, CD30 is strongly expressed by large EATL tumor cells (Sibon et al. ASH 2013). Based on their findings, Sibon et al. initiated a pilot study in 2012 to explore the combination of brentuximab vedotin and chemotherapy followed by ASCT as frontline treatment for EATL. Initially, IVE/MTX was the associated chemotherapy regimen, however after data presented at ASH 2012 (Fanale et al. ASH 2012), this was replaced with CHP. The findings are summarized below:

Based on the promising results of the pilot study, a phase 2 study is being planned to further explore brentuximab vedotin, CHP and ASCT as frontline therapy for patients with EATL.
Dr Visser concluded his talk by once more summarizing the available treatment options for EATL including surgery, CHOP[-like] and ASCT consolidation. However, he emphasized the important of prevention and effective treatment of RCD II. He also encouraged participation in trials exploring ASCT preceded by brentuximab vedotin-CHP and/or high-dose MTX as frontline therapy for EATL patients.

Understanding your specialty helps us to deliver the most relevant and engaging content.
Please spare a moment to share yours.
Please select or type your specialty
 Thank you
Thank youNewsletter
Subscribe to get the best content related to lymphoma & CLL delivered to your inbox








