All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a Healthcare Professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The Lymphoma Hub uses cookies on this website. They help us give you the best online experience. By continuing to use our website without changing your cookie settings, you agree to our use of cookies in accordance with our updated Cookie Policy
Introducing

Now you can personalise
your Lymphoma Hub experience!
Bookmark content to read later
Select your specific areas of interest
View content recommended for you
Find out moreThe Lymphoma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Lymphoma Hub cannot guarantee the accuracy of translated content. The Lymphoma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
iwCLL 2017 | PI3K Isoform Inhibitors in CLL: idelalisib, duvelisib, and TGR-1202
Bookmark this article
During iwCLL, on 15th May 2017, the “Additional Therapies for the Relapsed/Refractory CLL Patient” session took place and was co-chaired by Michael Keating (MD Anderson Cancer Center) and Jacqueline Barrientos (The Feinstein Institute for Medical Research).
During this session, Jennifer R. Brown, MD, PhD, from the Dana-Farber Cancer Institute, Boston, Massachusetts, USA, gave a presentation titled “Where Are We With PI3K Isoform Inhibitors in CLL?”
Idelalisib
Jennifer Brown began the talk by discussing idelalisib (GS-1101), a highly selective inhibitor of PI3Kδ.
In a phase I trial (NCT00710528, NCT01090414) idelalisib was evaluated in 54 patients with R/R CLL with adverse characteristics including bulky lymphadenopathy (80%), extensive prior therapy (median = 5; range, 2–14), treatment-refractory disease (70%), unmutated IGHV (91%), and del(17p) and/or TP53 mutations (24%). Six dose levels of oral idelalisib, ranging from 50 to 350mg once or twice daily, were investigated. Patients remained on continuous therapy while clinical benefit was assessed. Idelalisib inhibited PI3Kδ leading to reduced Akt phosphorylation in patients’ CLL cells and significantly reducing levels of serum chemokines. The most frequent ≥grade 3 AEs included pneumonia (20%), neutropenic fever (11%), and diarrhea (6%). Idelalisib achieved nodal responses in 81% of patients. The ORR was 72%, with 39% of patients meeting the criteria for PR as defined by the iwCLL 2008 criteria; 33% met the recently updated criteria of PR with treatment-induced lymphocytosis. Median PFS for all patients was 15.8 months. The findings of this phase I study highlight the clinical utility of idelalisib to inhibit PI3Kδ (Brown et al. 2014).
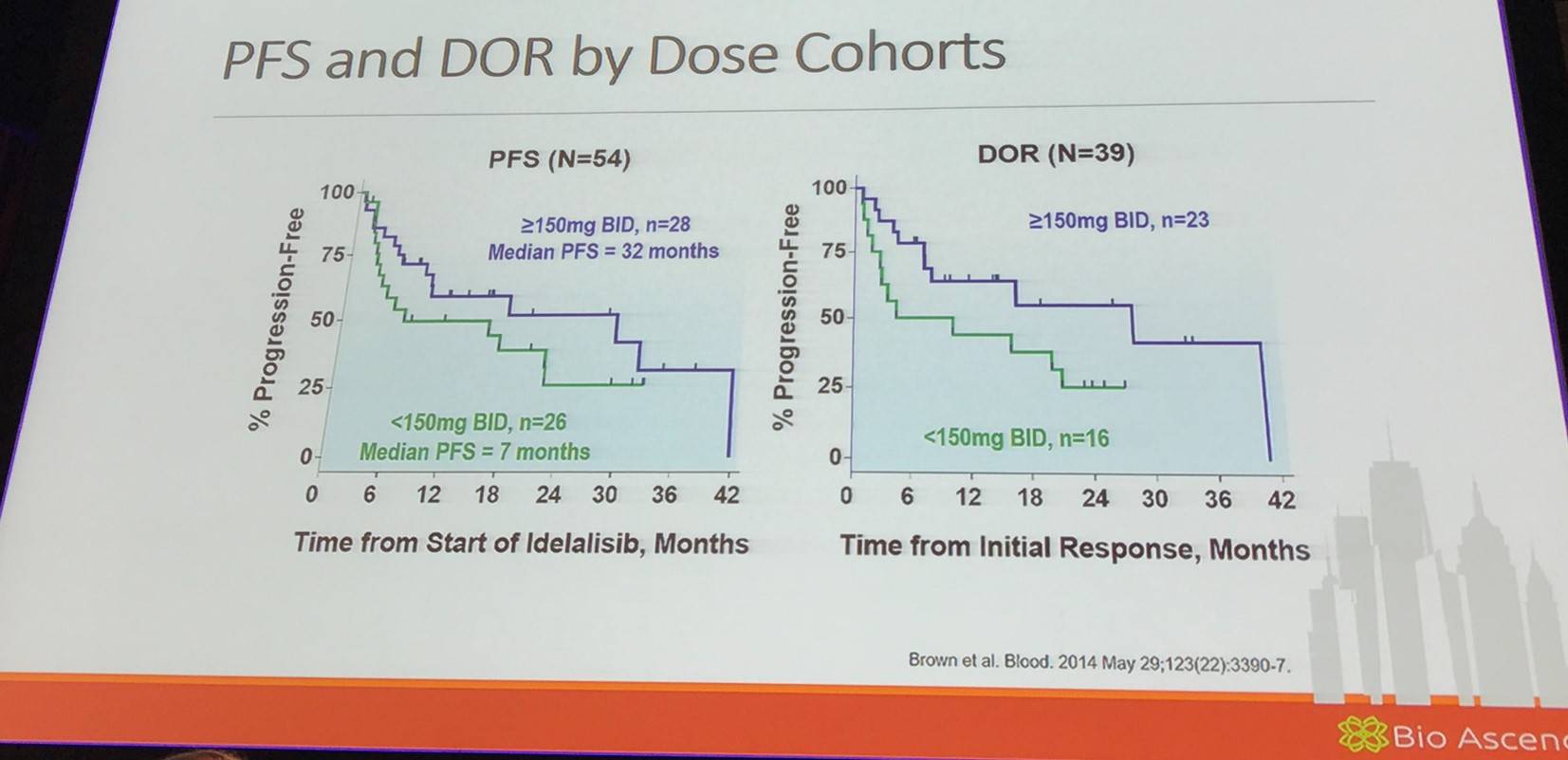
Jennifer Brown moved on to discuss Study 116 (NCT01539512); a phase III, randomized, double-blind, placebo-controlled study of idelalisib in combination with rituximab for previously treated CLL.
|
Eligibility criteria |
|
|---|---|
|
Relapsed CLL |
- CLL progression <24 months since last therapy - Treatment warranted according to iwCLL criteria |
|
Prior therapies |
- ≥1 anti-CD20 antibody containing therapy or ≥2 prior cytotoxic therapies |
|
Appropriate for non-cytotoxic therapy |
- CIRS score >6 or CrCl <60ml/min (≥30mL/min) or Grade 3–4 neutropenia or thrombocytopenia due to prior myelotoxicity |
|
Bone marrow function |
- Any grade anemia, neutropenia, or thrombocytopenia allowed |
|
Karnofsky score |
- ≥40 |
Overall, 220 patients (110 per arm) with a median age of 71 years (78% were 65 years or older) and a median number of 3 prior therapies (range, 1–12) were randomized. Del(17p)/TP53 mutation was found in 43% and 84% had unmutated IGHV. PFS, including extension study, was higher for idelalisib+R (19.4 mo; 95% CI, 16.6–NR) vs. placebo+R (7.3 mo; 95% CI, 5.5–8.5; HR, 0.25; P < 0.0001) (Sharman et al. 2014).
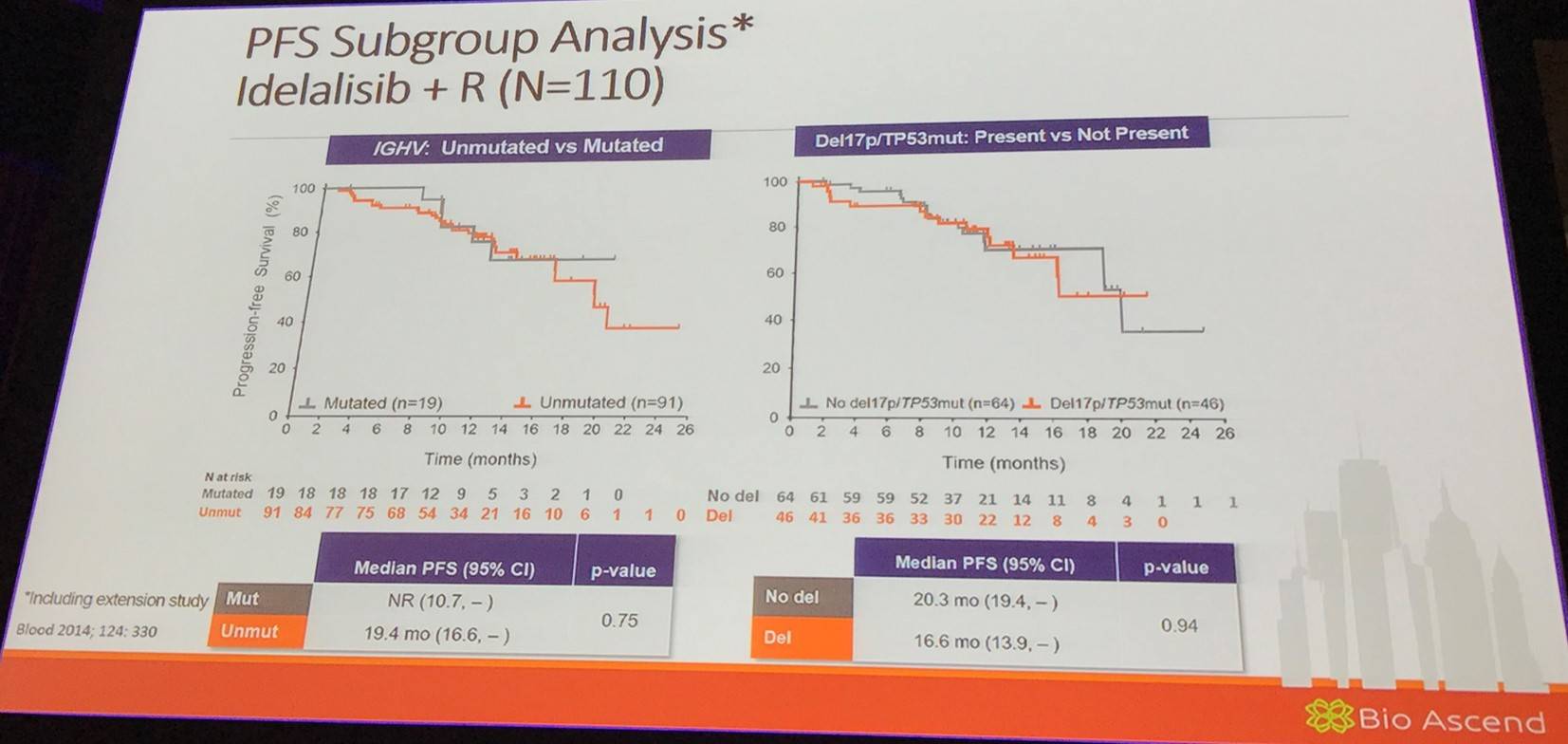
The talk then focused on results of a phase III, randomized, controlled, study assessing the efficacy and safety of idelalisib in combination with ofatumumab for previously treated CLL (Study 119; NCT01659021).
|
|
Arm A (IDELA/OFA) |
Arm B (OFA) |
HR/OR2 |
|---|---|---|---|
|
Med PFS, mo |
16.3 |
8.0 |
HR = 0.27; P < 0.0001 |
|
ORR, % |
75.3 |
18.4 |
OR = 15.9; P < 0.0001 |
|
LNR, % |
93.3 |
4.9 |
OR = 487; P < 0.0001 |
|
Med OS, mo |
20.9 |
19.4 |
HR = 0.74; P = 0.27 |
|
Med PFS: del(17p)/TP53 mut, mo |
13.7 |
5.8 |
HR = 0.33; P < 0.0001 |
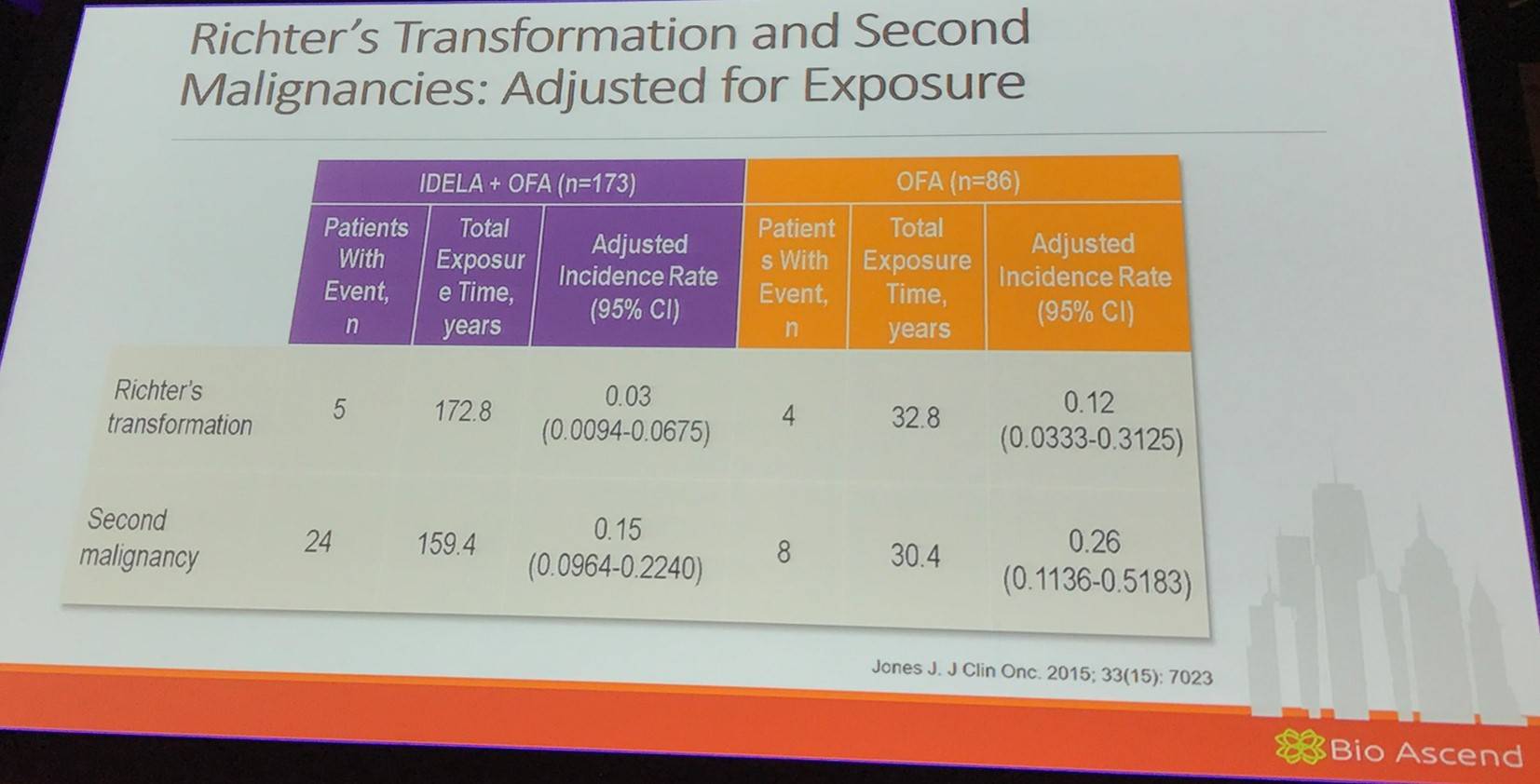
Additionally, results from Study 115, a phase III, randomized, double-blind trial of idelalisib combined with bendamustine and rituximab for previously treated CLL (NCT01569295) were presented. Median follow-up time was 12 months (Zelenetz et al. ASH 2015; LBA-5).
|
|
IDELA+BR |
BR+PBO |
|---|---|---|
|
Median PFS, mo |
23.1 |
11.1 |
|
HR (95% CI); p-value |
0.33 (0.24–0.45); P < 0.0001 |
|
Next, results from a phase II study of idelalisib plus rituximab in newly diagnosed older patients with CLL were discussed (NCT01203930). Median time on therapy was 22.4 months, with an ORR of 97% and a CR rate of 19%. PFS at 36 months was 83%. In the 9 patients with del(17p)/TP53 mutation, ORR was 100% with a CR rate of 33%.
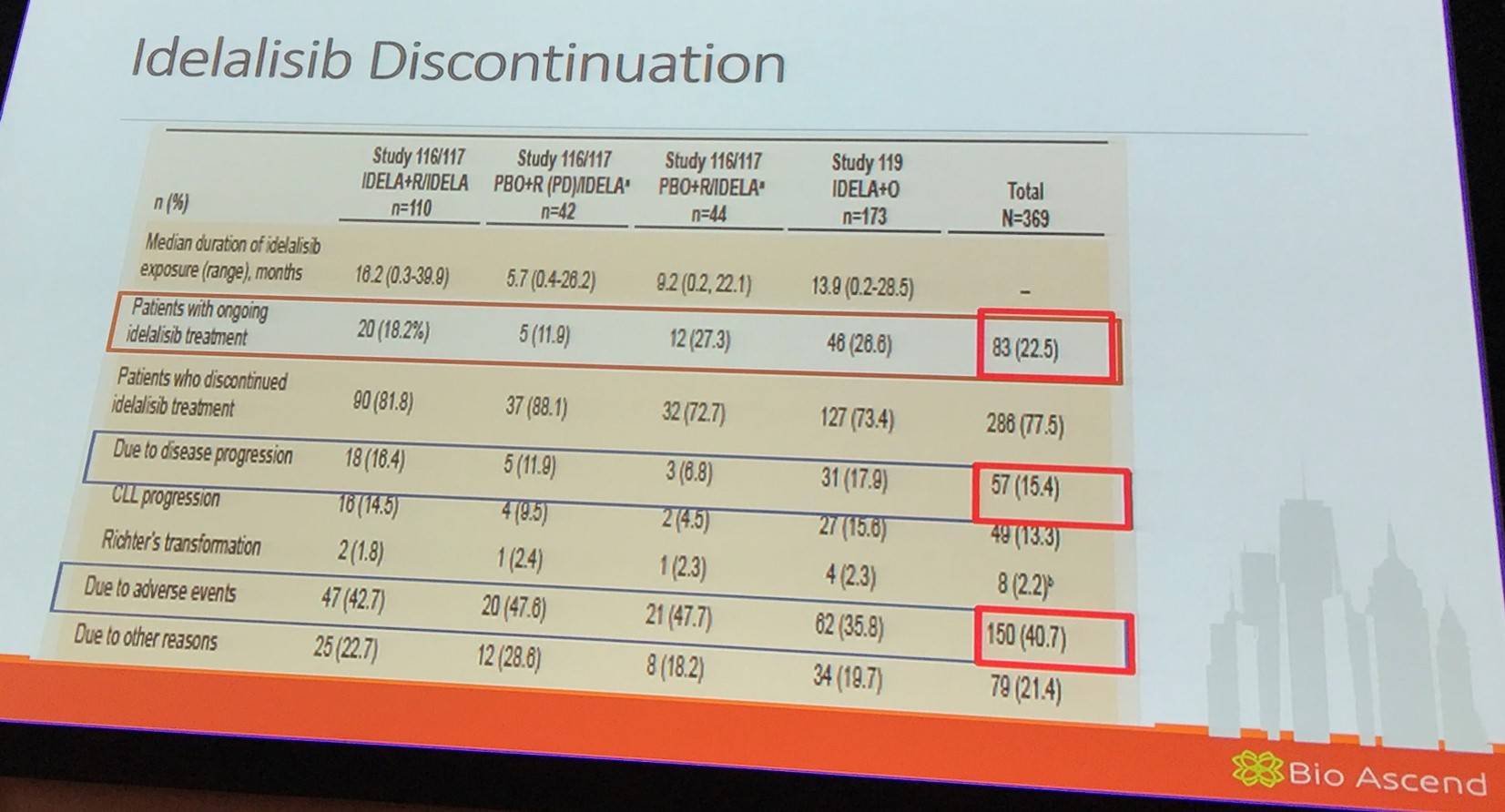
Jennifer Brown then highlighted discontinuation of idelalisib across Study 116/117 and Study 119.
Toxicity and the success of re-challenge after ibrutinib dose interruptions (Coutre S. et al. EHA 2015; S433):
|
Patients, n (%) |
N = 760 |
|---|---|
|
Grade ≥3 diarrhea/colitis |
106 (14%) |
|
Rechallenged |
71/106 (67%) |
|
Successful rechallenge |
41/71 (58%) |
|
Grade ≥3 ALT/AST elevation |
109 (14%) |
|
Rechallenged |
82/109 (75%) |
|
Successful rechallenge |
63/82 (77%) |
|
Grade ≥3 rash |
45 (6%) |
|
Rechallenged |
34/45 (76%) |
|
Successful rechallenge |
27/34 (79%) |
|
Any grade pneumonia |
24 (3%) |
|
Rechallenged |
13/24 (54%) |
|
Successful rechallenge |
9/13 (69%) |
In terms of managing idelalisib-related immune AEs, it is important to be vigilant regarding the onset of the classic pattern of toxicity; the risk is higher in less heavily pre-treated and younger patients. It was advised to hold the drug early in the event of significant transaminitis, diarrhea, or intestinal pneumonitis. Moreover, evaluate completely for infection. Start steroids if likely to be drug-related and not resolving.
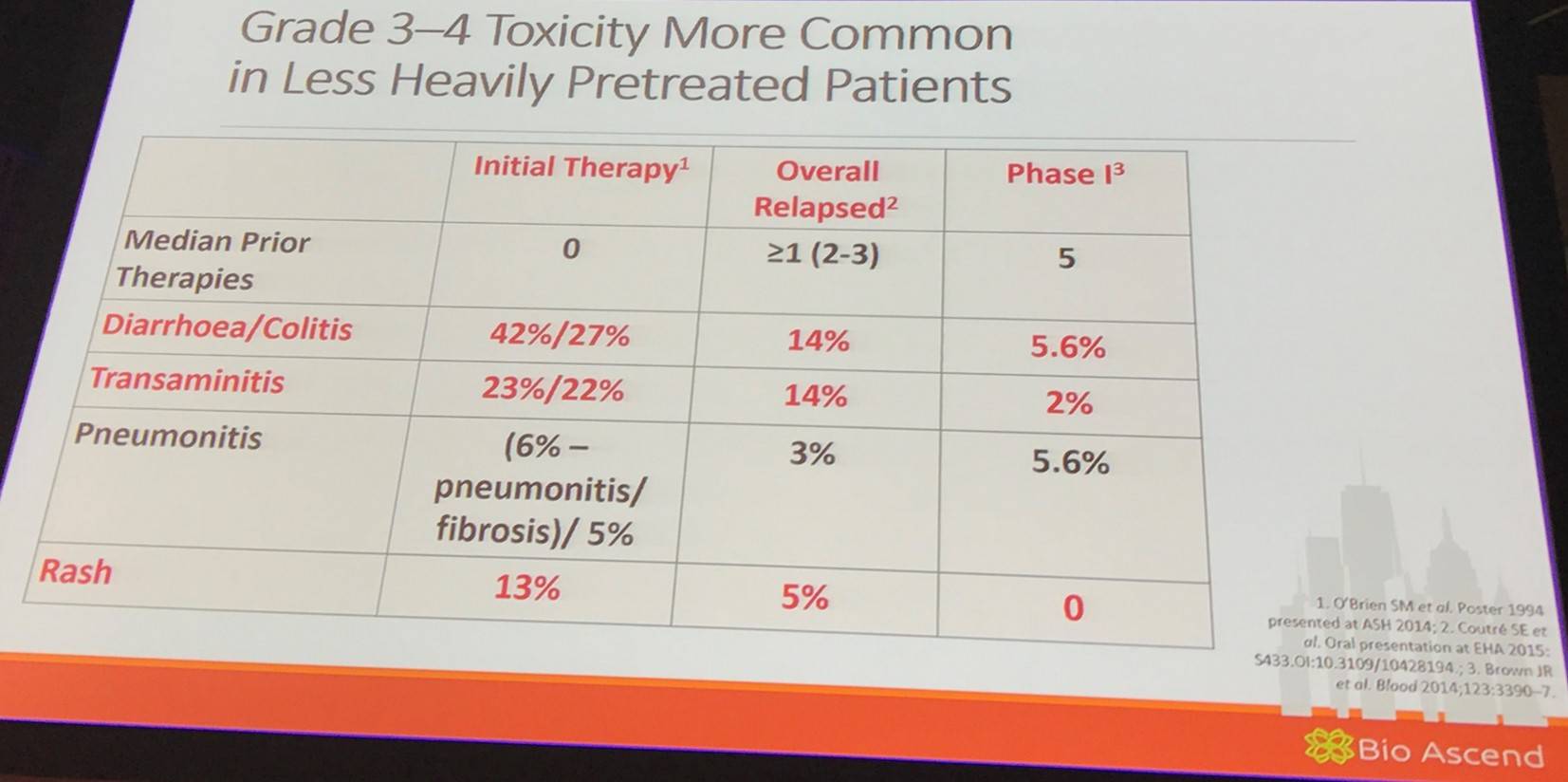
Jennifer Brown then discussed a phase II study (NCT02135133) of idelalisib plus ofatumumab in newly diagnosed CLL/SLL. At last analysis, 24 patients enrolled and median time on therapy was 7.7 months (range, 0.7–16.1 months). Median follow-up time was 14.7 months (range, 1.2–16.8 months). Hepatotoxicity was frequent and often severe, with 52% of patients experiencing ≥Grade 3 hepatotoxicity. A total of 79% (19 patients) experienced either ≥Grade 1 ALT or AST elevation during the study, and 54% (13 patients) experienced ≥Grade 3 transaminitis. Median time to development of transaminitis was 28 days, occurring before ofatumumab introduction. There was much evidence to suggest that this hepatotoxicity was immune mediated. A lymphocytic infiltrate was observed in liver biopsy specimens taken from 2 subjects with transaminitis, and levels of CCL-3 and CCL-4 (pro-inflammatory cytokines) were elevated in patients who experienced hepatotoxicity. All cases of transaminitis resolved either by holding the drug, administering immunosuppressants, or both; rates of recurrent toxicity were lower in patients administered with steroids upon idelalisib re-initiation. Decreased peripheral blood regulatory T-cells was observed in patients experiencing toxicity on therapy, consistent with an immune-mediated mechanisms. Younger age and mutated IGHV status were significant risk factors for the development of hepatotoxicity; 100% of patients 65 years old or younger required systemic steroids for toxicities (Lampson et al. 2016).
The connection between p110δ and Tregs was discussed. Mutations that disrupt Treg function in mice and humans leads to autoimmune syndromes with hepatitis, enteritis, and pneumonitis. Mice with kinase dead p110δ develop autoimmune colitis and have decreased numbers/function of regulatory T-cells.
Lampson et al. found that 11/15 patients (73%) with matched samples had a reduction in the percentage of T-cells over time after first-line therapy with idelalisib. The proportion of regulatory T-cells in the peripheral blood decreases on idelalisib therapy, providing a possible explanation of early hepatotoxicity.
Following this, Jennifer Brown gave an update on the currently reported safety information of idelalisib:
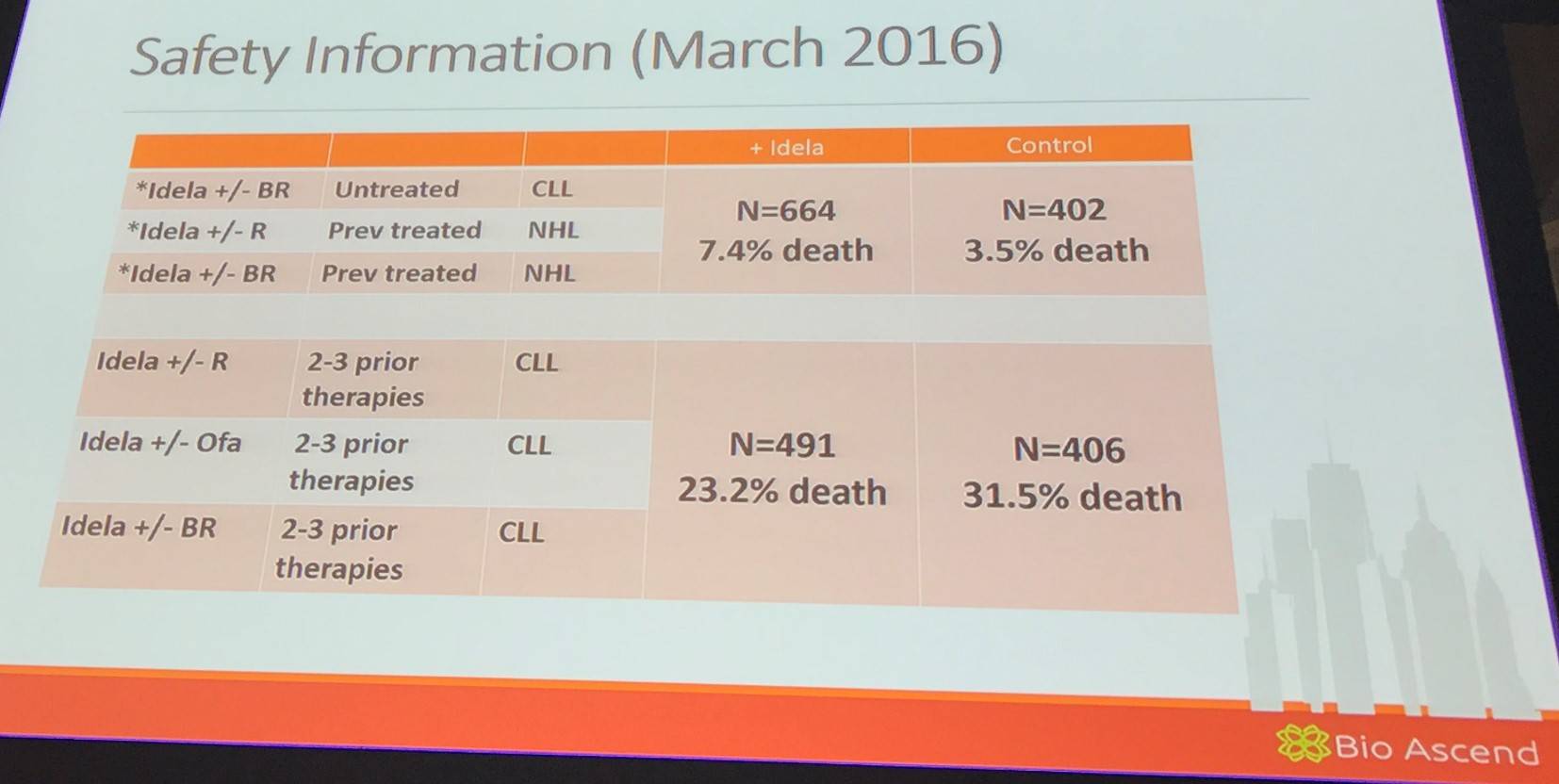
She then went on to explain that the majority of deaths in early idelalisib studies were mainly due to routine bacterial infection, neutropenia, or sepsis, and not opportunistic infections or autoimmune toxicity. Review of previous data highlights the risk of infection and neutropenia, even in first-line single-agent studies:
- Grade 3–4 neutropenia = 22–28%; febrile = 3–5%
- Grade 3–4 infections = 17%
- PJP / CMV cases on all studies

Duvelisib
The talk then focused on duvelisib (IPI-145), an oral PI3Kδ-γ inhibitor, which inhibits malignant B- and malignant T-cell survival. It has direct effects on tumor cells and disrupts tumor cell interactions within the microenvironment.
Jennifer Brown outlined results from the phase IPI-145-02 study (NCT01476657) of duvelisib in patients with advanced hematologic malignancies and also contained an expansion cohort of treatment naïve CLL patients.
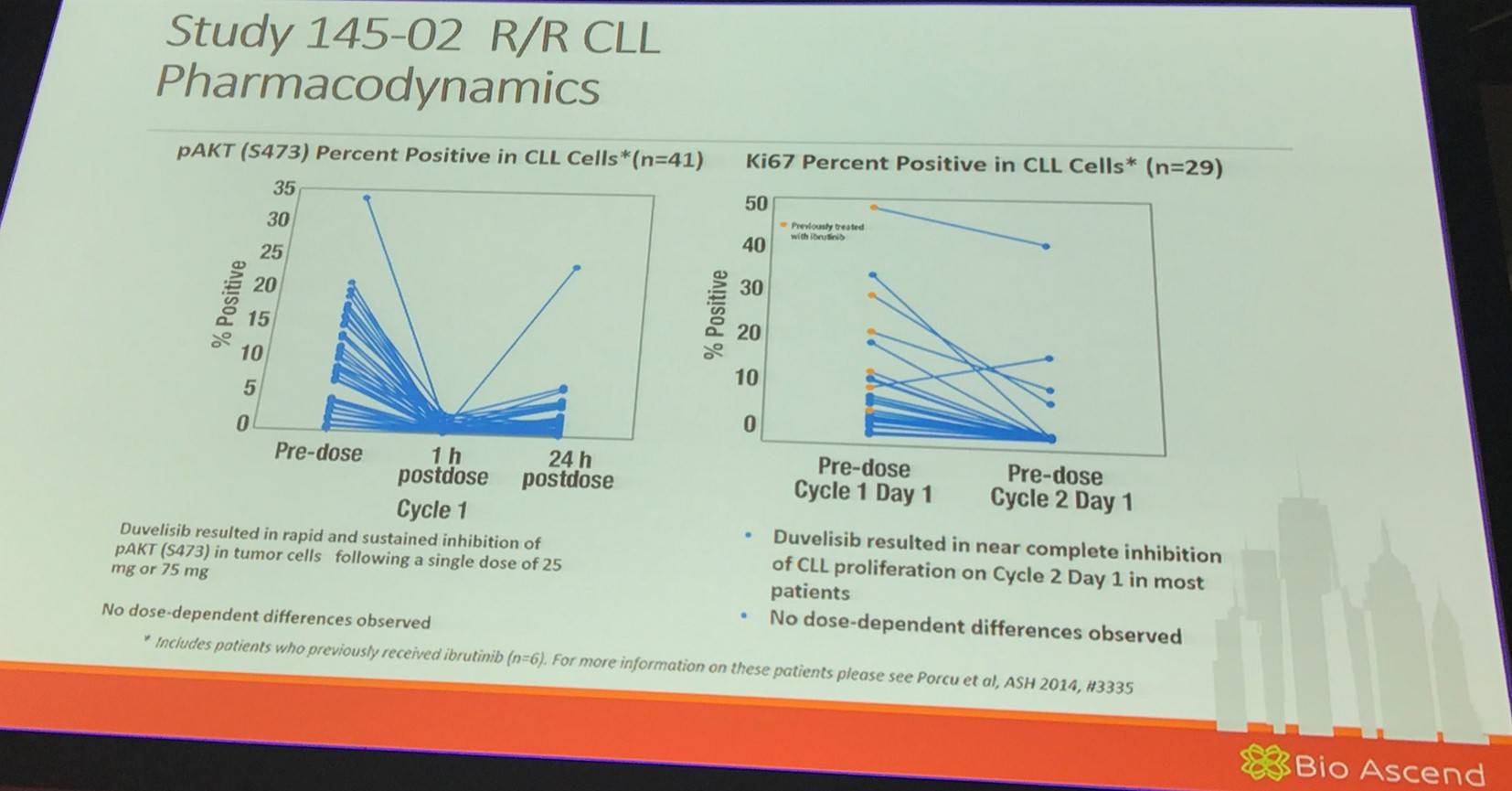
At the time of presentation at ASCO 2013 (abstract 7070), in the 16 patients with CLL (5 in the dose escalation <25 mg BID and 11 in a 25 mg BID EC), the median number of cycles received was 2.7 (range, 1–14) and 81% remained on study. TRAEs were experienced by 10 CLL patients (63%), similar to the incidence observed in the total study population (56%). The most common >Grade 3 TRAEs in CLL patients was neutropenia (25%). No >Grade 3 ALT elevations were reported in CLL patients. Among evaluable patients with CLL (n=11), 9 patients (82%) had a PR or nodal response after 2 cycles of IPI-145, with a best response of 6 PRs, 4 SD (all nodal responses), and 1 PD.
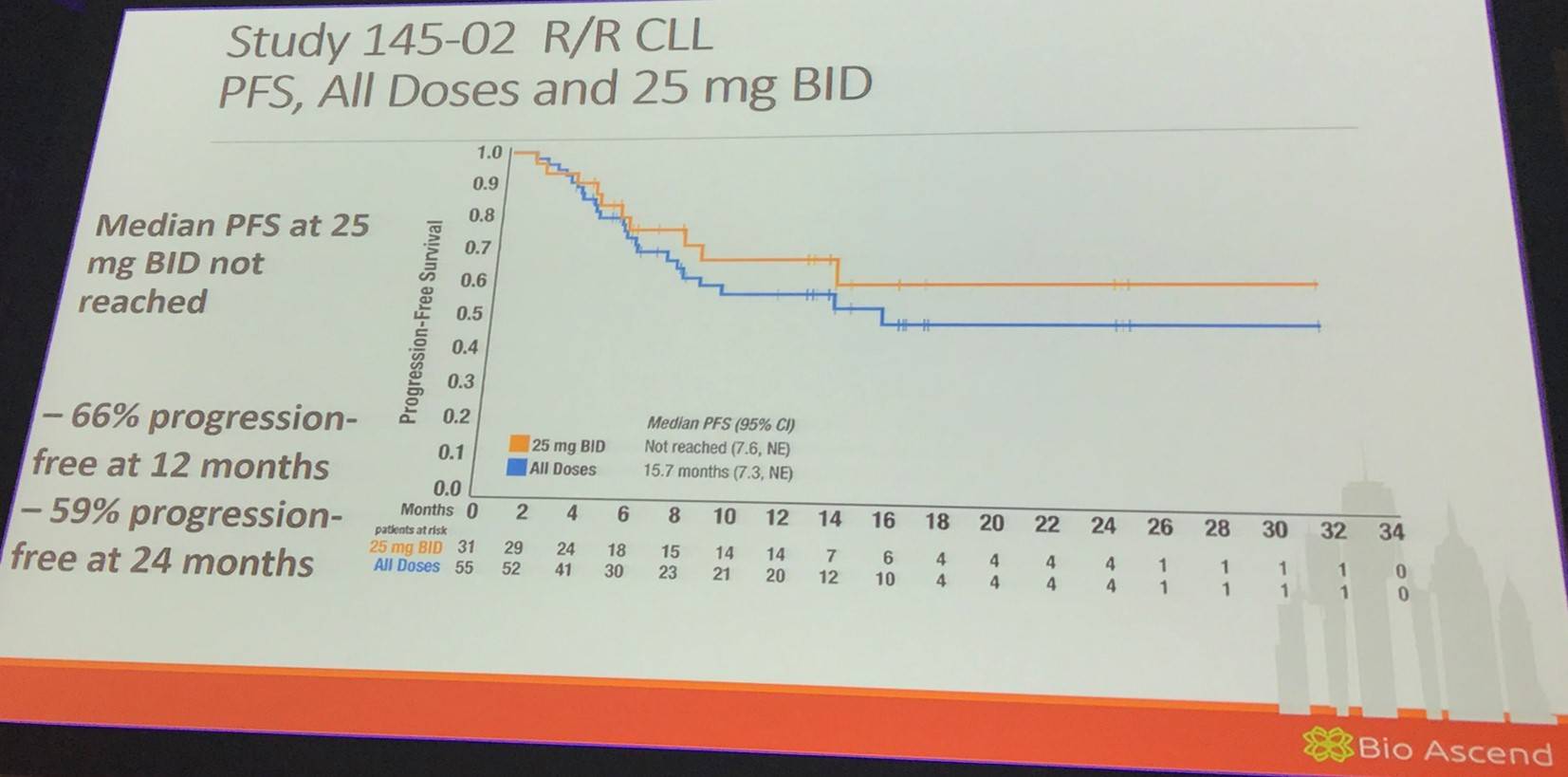
The talk then focused on duvelisib for the treatment of patients who had previously received ibrutinib:
|
Population |
n |
CR |
PR |
SD |
PD |
ORR |
|---|---|---|---|---|---|---|
|
CLL |
6 |
0 |
1 (17) |
5 (83) |
0 |
1 (17) |
|
aNHL |
5 |
0 |
2 (40) |
1 (20) |
2 (40) |
2 (40) |
|
Disposition |
CLL (N=6) |
aNHL (N=7) |
|---|---|---|
|
Median time on treatment, mo; n (range) |
3.8 (2.8–14.3) |
2.3 (0.5–9.0) |
|
Discontinued treatment |
5 (83) |
5 (71) |
|
Progressive disease |
4 (67) |
3 (43) |
TGR-1202
Jennifer Brown also discussed TGR-1202, a “next generation” PI3Kδ inhibitor. It has significant structural differences to other PI3Kδ inhibitors; EC50 for delta = 2.2nM in cell free system (Burris et al. ASCO 2015; 2513). The phase I TGR-1202-101 study, which aimed to assess the safety and efficacy of TGR-1202 in patients with R/R hematologic activities, was outlined:
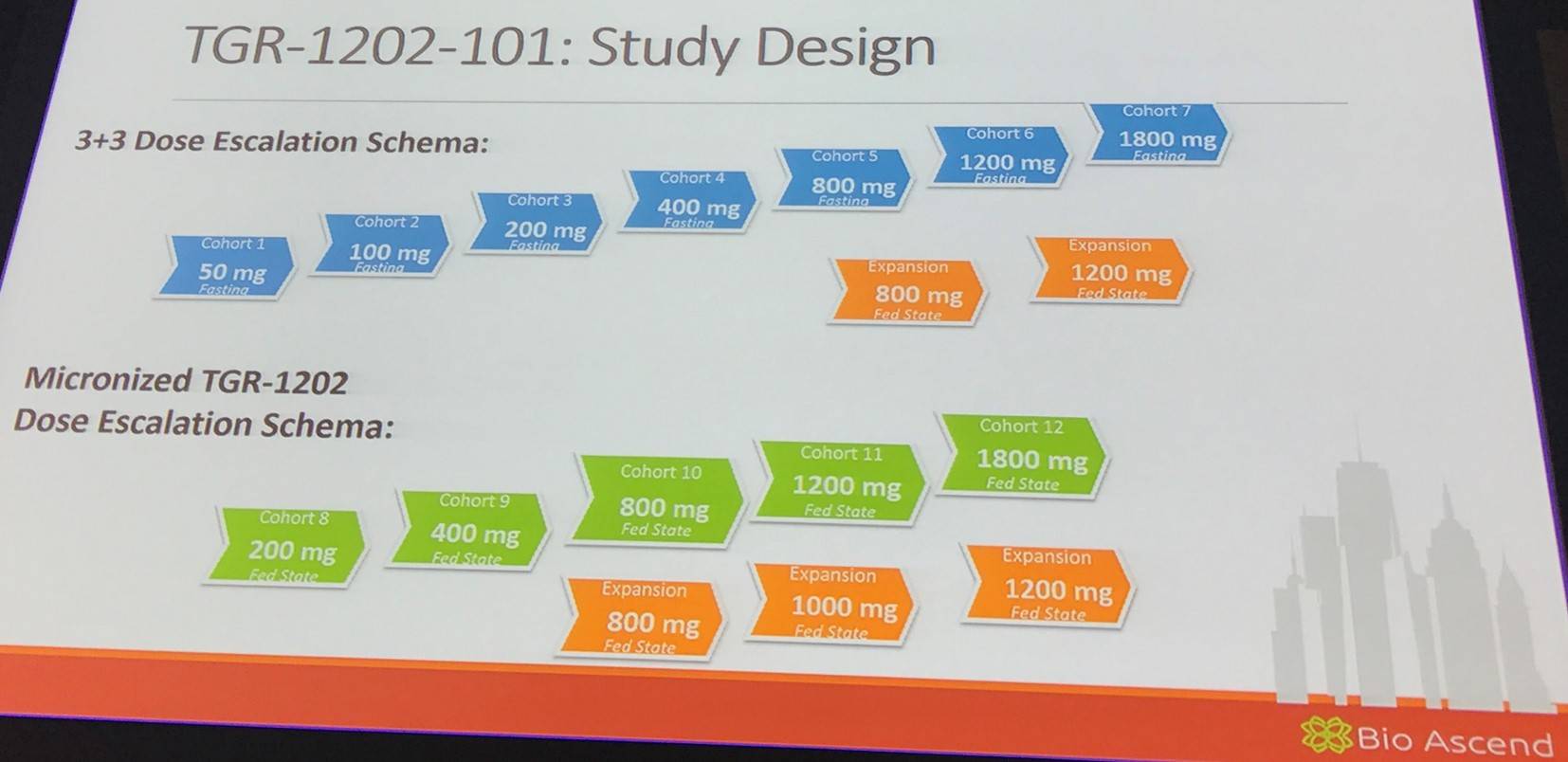
52 patients were exposed to the micronized formulation at dose levels ≥800mg with 27 patients for a duration in excess of 6 cycles. The longest patient on study received TGR-1202 for more than 34 cycles (more than 2.5 years). TGR-1202 was well tolerated with limited Grade 3–4 AEs; only 2% experienced Grade 3–4 increases in AST/ALT. Grade 3 pneumonia was reported in 5% (n=5). Of the 81 patients treated, no colitis has thus far been reported. TGR-1202 was stopped in 7% (n=6) for AEs (O’Connor et al. ASH 2015; 4154).
Jennifer Brown also presented data comparing the safety profile of TGR-1202 to other PI3K inhibitors:
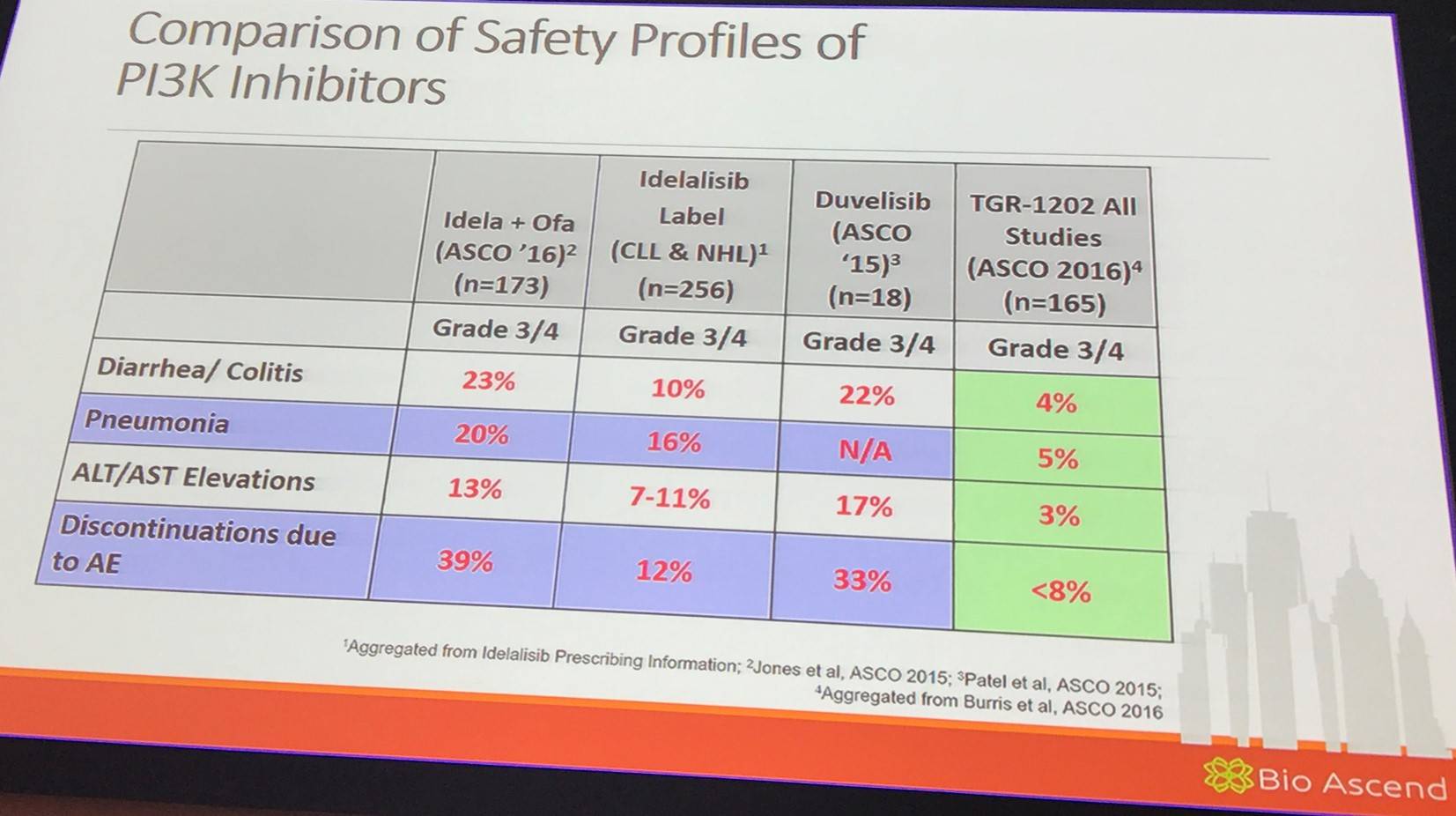
Lastly, it has been found that TGR-1202 is not only selective for PI3Kδ but also inhibits CK1-ε.
Conclusion
The talk was finished with a concise summary slide, as well as possible future applications:

PI3K inhibitors are highly active and are still much needed to treat B-cell malignancies. Possible settings for idelalisib include:
- Single-agent in patients intolerant to or relapsed after BTK inhibitors
- Identify a good biomarker for tolerance
- Alternative schedule or
- Identify a combination partner that might mitigate toxicity.
This immune activation represents an opportunity to use PI3Kδ inhibitors as anti-tumor immunomodulatory agents.
We interviewed Prof. Jennifer Brown at iwCLL 2017 about her presentation; click here to view.
- Brown J.R. Where Are We With PI3K Isoform Inhibitors in CLL? XVII International Workshop on Chronic Lymphocytic Leukemia; 2017 May 12–15; New York, USA.

Understanding your specialty helps us to deliver the most relevant and engaging content.
Please spare a moment to share yours.
Please select or type your specialty
 Thank you
Thank youRelated articles
Newsletter
Subscribe to get the best content related to lymphoma & CLL delivered to your inbox








