All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a Healthcare Professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The Lymphoma Hub uses cookies on this website. They help us give you the best online experience. By continuing to use our website without changing your cookie settings, you agree to our use of cookies in accordance with our updated Cookie Policy
Introducing

Now you can personalise
your Lymphoma Hub experience!
Bookmark content to read later
Select your specific areas of interest
View content recommended for you
Find out moreThe Lymphoma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Lymphoma Hub cannot guarantee the accuracy of translated content. The Lymphoma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
ICML 2017 | Response adapted therapies for Hodgkin Lymphoma – Meet the Professor Session
Bookmark this article
On the 14th June 2017, during the 14th International Conference on Malignant Lymphoma (ICML) in Lugano, Switzerland, the LH attended a session titled “Response-adapted therapy in Hodgkin Lymphoma” that was presented by Peter Johnson, MA, MD, FRCP, from the University of Southampton, Southampton, UK.
Johnson began by emphasizing that, despite incidence of Hodgkin Lymphoma (HL) remaining stable, mortality has steadily decreased over the last 3 decades. It is estimated that 80% of HL patients are cured with first-line therapy (90% in early-stage disease, 75% in advanced disease), but this is an underestimate of success. Over 80% of patients are also alive at 10 years and so the main priorities of research are to try and improve this percentage even further and to minimize toxicity.
The median age of patients is only 30–35 years with “a lot of life ahead of them”. There are a number of late effects that we need to try to avoid as the cure rate increase:
- Secondary MDS/AML from alkylating agents
- Solid tumors from extended field radiation
- Pulmonary fibrosis from bleomycin
- Ischemic heart disease from mediastinal irradiation and doxorubicin
- Infertility from alkylating agents
Moreover, Johnson noted that the risk of coronary heart disease remains a challenge even at low doses of radiotherapy; there has been a substantial increase in cumulative incidence (van Nimwegen et al. JCO. 2016). It appears that the “dilemma of therapy” is balancing cure and morbidity; as one goes up, the other goes up even higher.
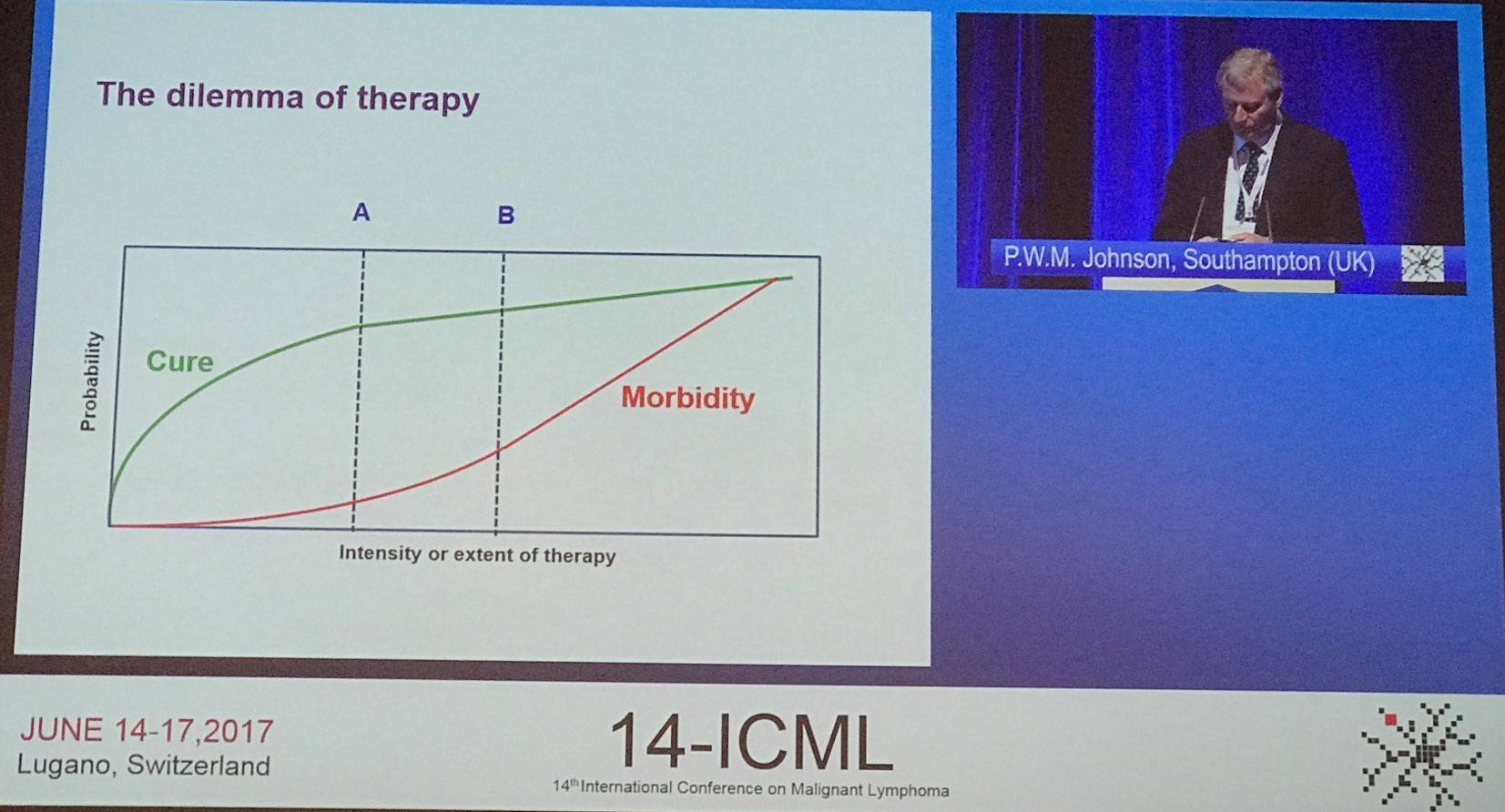
It was asked if we have to administer toxic treatment to the more than 80% of patients who can be cured with something less harmful? For each patient avoiding recurrent disease after more treatment, 15–20 may be needlessly put at risk of long-term toxicities. Currently, most deaths are caused by things of than lymphoma: disease control and survival are not one and the same. We are in the era for personalized medicine, so how can we implement it?
We need to be able to distinguish worse from “less bad” disease. Johnson stated that we are increasingly looking at baseline PET to help us identify baseline clinical features. He eluded to results which will be presented at ICML in abstract #19: in advanced HL, baseline Total Lesion Glycolysis (TLG) and Metabolic Tumor Volume (MTV) were significantly associated with PET-CT after 2 ABVD (PET2) response. A strong, independent risk factor for prognosis was found to be TLG, which might potentially aid selection of patients most likely to benefit from more intensive earlier therapy. A multivariate model including TLG may evaluate risk better than current clinical parameters; however, further work is needed to validate this. A model like this one could be of particular use in patients with negative PET2 scans, who achieved a 3-year PFS much lower than expected (85%).
In terms of baseline biological assessments, Scott et al. (JCO. 2016) devised a Gene Expression Profiler (GEP)-based, 23-gene outcome predictor model for OS, developed in formalin-fixed paraffin-embedded tissue biopsies from 290 patients enrolled onto the E2496 Intergroup trial (NCT00003389). The model was found to identify patients with advanced-stage classical HL (cHL) at a higher risk of death when administered standard-intensity up-front treatment approaches. However, Burton et al., whose findings will be presented on Thursday 15th June (abstract #81), found that this model was not predictive of PET2 response and is not an acceptable strategy for informing treatment escalation.
Currently, the best way we have of distinguishing worse from “less bad” disease is by assessing the response to therapy. Numerous studies have found that negative early interim FDG-PET is a strong and independent predictor of good outcomes in HL patients, and positive scans are highly predictive of progression in patients with advanced-stage or extranodal disease. However, is FDG-PET reliable and reproducible? Johnson answered this concern by stating that the Deauville 5-point scoring system has been proved to be reproducible (Barrington et al. EJNMMI. 2010), which is surprising considering it is a visual score. A high level of concordance has been found between PET experts and it is easy to apply.
Randomized trials testing the role of interim PET in early-stage disease were then discussed:
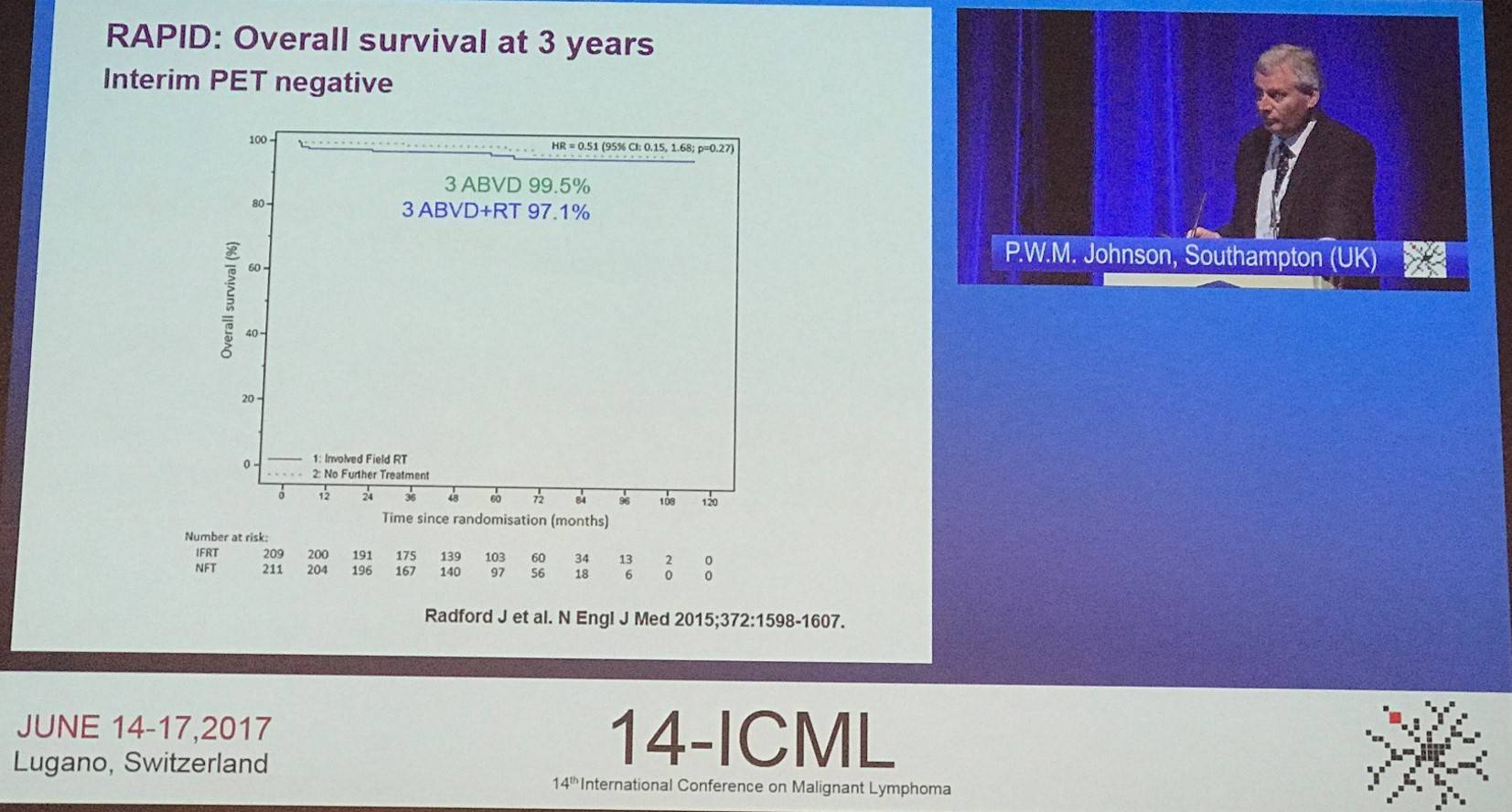
The phase III, randomized RAPID trial (NCT00943423) recruited 602 newly diagnosed stage IA or IIA HL patients, and 571 of these underwent PET scanning. PET-negativity was determined in 426 of these patients (74.6%), 420 of whom were randomized to radiotherapy group (n=209) or no further therapy (n=211). At a median follow-up of 60 months, 8 instances of PD had occurred in the radiotherapy group and 8 patients had died (3 with PD, 1 from HL). In the no further treatment group, there had been 20 instances of PD and 4 patients had died (2 with PD; none due to HL). 3-year PFS rate was 94.6% (95% CI, 91.5–97.7) in the radiotherapy group and 90.8% (95% CI, 86.9–94.8) in the no further treatment group, with an absolute risk difference of -3.8 percentage points (95% CI, -8.8 to 1.3). Patients in this study with early-stage HL and PET-negative findings after 3 cycles of ABVD had very good outcomes either with or without consolidative radiotherapy (Radford et al. N Engl J Med. 2015).
H10, a phase III, randomized trial by EORTC/GELA/FIL assessed early FDG-PET scan guided treatment adaptation compared to standard combined treatment in patients with favorable or unfavorable supradiaphragmatic stage I–II HL. Of 1,950 randomized patients, 1,925 received an early PET; of these, 361 patients (18.8%) were positive. In positive patients, 5-year PFS improved from 77.4% for standard ABVD followed by Involved Node Radiotherapy (INRT) to 90.6% for intensification to BEACOPPesc + INRT (HR, 0.42; 95% CI, 0.23–0.74; P = 0.002). In PET-negative patients, 5-year PFS rates in the favorable group were 99.0% for ABVD + INRT versus 87.1% for ABVD alone (HR, 15.8; 95% CI, 3.8–66.1) in favor of ABVD + INRT; in the unfavorable group, this was 92.1% versus 89.6% (HR, 1.45; 95% CI, 0.8–2.5) in favor of ABVD + INRT. This study concluded by stating that in stage I–II HL, early PET-positive response can accurately inform treatment escalation. In PET-negative patients, risk of relapse was higher when INRT was omitted, particularly in favorable patients.
The role of interim PET has also been assessed in patients with advanced disease in trials such as RATHL (updated results will be presented during ICML by Judith Trotman; abstract #54), the GITIL 0607 trial, and the LYSA AHL2011 trial.

In the phase III RATHL study (NCT00678327), the associations between baseline factors and outcomes in interim PET-negative patients are outlined below:
|
|
|
PFS HR (95% CI) |
P |
3-year PFS % |
OS HR (95% CI) |
P |
3-year OS % |
|---|---|---|---|---|---|---|---|
|
Stage |
II |
1.00 |
<0.001 |
89.2 |
1.00 |
0.002 |
99.2 |
|
III |
1.9 (1.3–2.8) |
83.1 |
4.2 (1.7–10.6) |
96.9 |
|||
|
IV |
2.0 (1.3–3.0) |
79.7 |
4.3 (1.7–11.1) |
95.3 |
|||
|
IPS |
0–2 |
1.00 |
0.027 |
86.7 |
1.00 |
<0.001 |
98.6 |
|
≥3 |
1.4 (1.0–1.9) |
81.6 |
2.8 (1.5–5.3) |
95.2 |
|||
|
Bulk |
- |
1.00 |
0.18 |
83.4 |
1.00 |
0.12 |
97.2 |
|
+ |
0.8 (0.5–1.2) |
87.4 |
0.5 (0.3–1.2) |
97.8 |
|||
|
PET2 |
1 |
1.00 |
0.65 |
85.2 |
1.00 |
0.41 |
95.1 |
|
2 |
0.9 (0.5–1.4) |
85.0 |
0.6 (0.2–1.3) |
98.1 |
|||
|
3 |
1.1 (0.6–1.7) |
84.2 |
0.6 (0.3–1.5) |
97.2 |
Johnson then summarized data from all the so far mentioned trials of control of lymphoma after treatment de-escalation in PET-negative patients; OS so far has not been found to be affected by de-escalation is PET-negative patients:
|
|
PFS |
OS |
|---|---|---|
|
RAPID |
3-year: 94.6% vs. 90.8% (P = 0.16) |
3-year: 97.1% vs. 99.5% |
|
H10 |
5-year: 99.0% vs. 87.1% |
5-year: 96.7% vs. 98.3% |
|
RATHL |
3-year: 85.4% vs. 84.0% (P = 0.62) |
3-year: 97.2% vs. 97.6% |
|
LYSA |
2-year: 91.6% vs. 88.3% (P = 0.79) |
2-year: 99% vs. 98% |
But, what about escalation after PET-positive scans? There are relatively few trials in this area, only the H10 trial has demonstrated that escalation is effective (5-year PFS with ABVD + INRT was 77.4% vs. 90.6% with BEACOPPesc + INRT, as mentioned above). The RATHL study did not get patient group backing for randomization.
Johnson also mentioned results from the phase II SWOG S0816 study, which assessed response-adapted therapy based on early FDG-PET for stage III–IV cHL. 2-year PFS in PET-negative and PET-positive patients was 82% and 64%, respectively (Press et al. JCO. 2016).
Recent results of the phase III HD18 trial by the German Hodgkin Study Group (NCT00515554) were also discussed; this trial aimed to assess whether intensifying BEACOPPescalated by adding rituximab would improve PFS in PET-positive patients after 2 courses of chemotherapy. 3-year PFS in patients administered BEACOPPescalated and R-BEACOPPescalated was 93.0% and 91.4%, respectively, and 3-year OS was 96.5% and 94.4%, respectively. The group concluded that the addition of rituximab to BEACOPPescalated did not improve the PFS of PET2-positive patients with advanced stage HL.
Johnson began rounding up the session by stating that FDG-PET provides an opportunity to tailor therapy to each individual patient, in order to achieve the best possible balance between anti-lymphoma effect and tolerability. Interim PET accuracy is influenced by the extent of the disease and the original treatment that is administered. All evidence to-date indicates that therapy de-escalation appears to retain efficacy as well as reduces morbidity. Additionally, exploration into therapy escalation has so far found it improves disease control and potentially improves mortality. Moreover, the application of interim PET means novel treatment strategies can be tested in patients with high-risk disease have achieved disappointing responses to currently available therapy.
It was asked if we can do better, and the answer is probably yes. Even when achieving negative interim PET disease, results in patients with more advanced disease could be improved. The performance of interim PET may potentially be improved with the use of biomarkers such as baseline TLG/MTV. Moreover, we should still actively look for novel forms of upfront therapy as well as improved therapeutic strategies for interim PET-positive patients.

Lastly, when comparing different initial approaches for advanced HL, larger amounts of BEACOPP received increases the chance of infertility (RATHL vs. LYSA AHL2011). Johnson also felt that, based on the LYSA data, interim PET is more accurate for stage 4 disease whereas interim PET performed better for stage II or III disease in the RATHL trial.
- Johnson P.W.M. Response-adapted therapy in Hodgkin Lymphoma 14th International Conference on Malignant Lymphoma; 2017 June 14–17; Lugano, Switzerland.

Understanding your specialty helps us to deliver the most relevant and engaging content.
Please spare a moment to share yours.
Please select or type your specialty
 Thank you
Thank youRelated articles
Newsletter
Subscribe to get the best content related to lymphoma & CLL delivered to your inbox








